Acropyga inezae
| Acropyga inezae | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Formicinae |
| Tribe: | Plagiolepidini |
| Genus: | Acropyga |
| Species group: | butteli |
| Species: | A. inezae |
| Binomial name | |
| Acropyga inezae Forel, 1912 | |
Nothing is known about the biology of this species.
Identification
LaPolla (2004): A member of the Acropyga butteli species group. Worker: 11 segmented antennae; mandible with 4 teeth, basal tooth enlarged and rectangular, apically truncated. Queen: As in worker with modifications expected for caste. Male: 12 segmented antennae; 4 mandibular teeth; parameres thin; digiti with truncated apices; cuspi meet digiti about half way along length of digiti. Compare with Acropyga butteli and Acropyga nipponensis.
This species can be recognized from other species in the butteli species-group since it only has 4 mandibular teeth as opposed to 5. The 3rd tooth from apical has a distinctive appearance that can be diagnostic; this tooth in some specimens examined looks similar in appearance to the truncated basal tooth. It seems likely that the unique shape of the 3rd tooth resulted from the fusion of 2 formerly separate teeth, but what is not clear is if A. inezae ever has 5 teeth, with the 3rd tooth being separated into 2 distinct teeth. The specimen sample available for study was small, so it cannot be completely discounted that 5 teeth may be possible. Future analysis of the butteli species-group should take the possibility of variation in teeth number into account for this species.
Keys including this Species
Distribution
Distribution based on Regional Taxon Lists
Indo-Australian Region: Borneo, Indonesia (type locality), Malaysia, Singapore.
Known only from Sumatra and Borneo.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
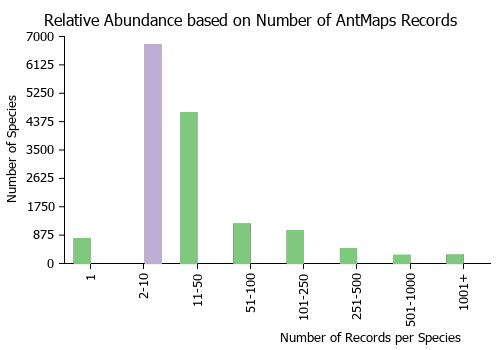
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
|
Biology
|
Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- inezae. Acropyga (Atopodon) inezae Forel, 1912m: 772 (w.) INDONESIA (Sumatra).
- [Also described as new by Forel, 1913k: 102.]
- Karavaiev, 1933a: 314 (q.m.).
- Status as species: Emery, 1925b: 30; Stärcke, 1930: 376 (in key); Karavaiev, 1933a: 312; Chapman & Capco, 1951: 212; Bolton, 1995b: 57; LaPolla, 2004a: 38 (redescription); Pfeiffer, et al. 2011: 36.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
LaPolla (2004) - (n=6): TL: 1.48-1.7; HW: 0.403-0.432; HL: 0.485-0.522; SL: 0.313-0.341; ML: 0.31-0.449; GL: 0.612-0.811; CI: 79.01-86.21; SI: 73.30-81.38.
Head: yellow; covered in layer of appressed hairs; head longer than broad; posterior margin entire; 11 segmented, incrassate antennae; scapes fail to reach posterior margin by about length of 1st three funicular segments; clypeus broad, convex medially; median portion clypeal anterior margin with rounded bulge giving clypeus a beak-like appearance; mandible broad with 4 teeth; basal tooth enlarged and rectangular, apically truncated; 3rd tooth without well-defined cusp and larger than 1 st and 2nd teeth, occasionally forming a truncated tooth similar to basal tooth; basal tooth offset from other teeth; anterior clypeal margin and inner mandibular margin nearly parallel. Mesosoma: yellow; pronotum covered with appressed hairs, long, erect hairs placed posteriorly; mesonotum flat, at about same height as propodeum; dorsum covered in appressed hairs; metanotal area indistinct; propodeum flat rounded into steep declivity. Gaster: yellow, covered in a thick layer of appressed hairs with scattered erect hairs throughout.
Queen
LaPolla (2004) - (n=2): TL: 2.74-2.92; HW: 0.52-0.526; HL: 0.571-0.583; SL: 0.412-0.439; ML:0.819-0.848; GL: 1.34-1.5; CI: 89.19-92. 12; SI: 60.22-72.78. As in worker caste with modifications expected for caste and with the following differences: scape slightly longer in queens, occasionally reaching posterior margin to slightly failing to reach posterior margin; color yellow to brownish-yellow.
Male
LaPolla (2004) - (n=2): TL: 1.66-1.78; HW: 0.316-0.362; HL: 0.364-0.38; SL: 0.218-0.23; ML: 0.464-0.557; GL: 0.831-0.844; CI: 86.81-95.26; SI: 60.22-72.78.
Head: brownish-yellow, darker toward apex around three prominent ocelli; head slightly longer than broad; eyes large, breaking outline of head in full frontal view; 12 segmented antennae; scapes fail to reach. posterior margin by about length of pedicel; clypeus broad, slightly convex medially, with erect hairs scattered throughout; shape of anterior clypeal margin similar to that of worker caste, but median tooth less defined; mandible broad with 4 teeth, basal tooth enlarged and rectangular, apically truncated as in worker caste. Mesosoma: yellow; pronotum short and collar-like, overarched by mesonotum; mesonotum rounded anteriorly; dorsum flat with erect hairs scattered throughout, especially on scutellum; propodeum short; declivity steep. Gaster: yellow; covered in a layer of appressed hairs, with scattered erect hairs throughout. Genitalia: in lateral view, parameres thin, taper to rounded apices; hairs evenly distributed across dorsal surface of parameres; in dorsal view parameres give genitalia a roughly triangular appearance; cuspi short, bent toward digiti, with peg-like teeth at apex; digiti longer than cuspi and bent toward cuspi with truncated apex; peg-like teeth found at about mid-length of digiti where cuspi meet.
Type Material
LaPolla (2004) - Acropyga (Atopodon) inezae Forel, 1912c: 772 (w.). Syntype worker(s?), INDONESIA: Sumatra (Naturhistorisches Museum, Basel) [examined). Lectotype is a worker labeled JSL TYPE # 113 is deposited at NHMB. Karavaiev, 1933: 314, description of queen and male.
References
- Forel, A. 1912n. Descriptions provisoires de genres, sous-genres, et espèces de Formicides des Indes orientales. Rev. Suisse Zool. 20: 761-774 (page 772, worker described)
- Forel, A. 1913l. Wissenschaftliche Ergebnisse einer Forschungsreise nach Ostindien ausgeführt im Auftrage der Kgl. Preuss. Akademie der Wissenschaften zu Berlin von H. v. Buttel-Reepen. II. Ameisen aus Sumatra, Java, Malacca und Ceylon. Gesammelt von Her (page 102, also described as new)
- Karavaiev, V. 1933a [1932]. Ameisen aus dem Indo-Australischen Gebiet, VII. Konowia 11: 305-320 (page 314, queen, male described)
- LaPolla, J.S. 2004a. Acropyga of the world. Contributions of the American Entomological Institute. 33(3):1-130. (page 38, worker, queen, male described)
- Wang, W.Y., Soh, E.J.Y., Yong, G.W.J., Wong, M.K.L., Benoit Guénard, Economo, E.P., Yamane, S. 2022. Remarkable diversity in a little red dot: a comprehensive checklist of known ant species in Singapore (Hymenoptera: Formicidae) with notes on ecology and taxonomy. Asian Myrmecology 15: e015006 (doi:10.20362/am.015006).
References based on Global Ant Biodiversity Informatics
- Chapman, J. W., and Capco, S. R. 1951. Check list of the ants (Hymenoptera: Formicidae) of Asia. Monogr. Inst. Sci. Technol. Manila 1: 1-327
- Forel A. 1912. Descriptions provisoires de genres, sous-genres, et espèces de Formicides des Indes orientales. Rev. Suisse Zool. 20: 761-774.
- Forel A. 1912. Descriptions provisoires de genres, sous-genres, et espèces de Formicides des Indes orientales. Revue Suisse de Zoologie 20: 761-774.
- Hashimoto Y., and M. Mohamed. 2011. Ground-dwelling ant diversity in Maliau Basin, Borneo: evaluation of hand-sorting methods to estimate ant diversity. Tropics 19(2): 85-92.
- Karavaiev V. 1933. Ameisen aus dem Indo-Australischen Gebiet, VII. Konowia 11: 305-320.
- LaPolla J.S. 2004. Acropyga (Hymenoptera: Formicidae) of the world. Contributions of the American Entomological Institute 33(3): 1-130.
- Pfeiffer M.; Mezger, D.; Hosoishi, S.; Bakhtiar, E. Y.; Kohout, R. J. 2011. The Formicidae of Borneo (Insecta: Hymenoptera): a preliminary species list. Asian Myrmecology 4:9-58

