Aenictus fuchuanensis
| Aenictus fuchuanensis | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Dorylinae |
| Genus: | Aenictus |
| Species: | A. fuchuanensis |
| Binomial name | |
| Aenictus fuchuanensis Zhou, 2001 | |
The species was collected in an experimental tree plantation (see Bruelheide et al. 2014) that was planted four years prior and at the time of collection still had an open character with a maximum tree height of 3 m and abundant patches of bare soil. Hence, A. fuchuanensis may be able to inhabit more open landscapes and not be restricted to forests, which may explain the relatively wide distribution of the species, which occurs from south Thailand to south-east China. (Staab 2015)
Identification
A member of the ceylonicus group. Jaitrong and Yamane (2013) – Aenictus fuchuanensis is similar to Aenictus maneerati, Aenictus pinkaewi, and Aenictus sundalandensis in having the long cylindrical petiole and the short head (CI 100-112). It is most similar to A. pinkaewi but the sculpturing on the mesosoma is weaker than in the latter (anterior portion of pronotum densely punctate, lateral face of pronotum finely reticulate in the former, while promesonotum entirely punctate in the latter).
Keys including this Species
- Key to Aenictus ceylonicus group species of China
- Key to southeastern Asian Aenictus ceylonicus group species
Distribution
Distribution based on Regional Taxon Lists
Oriental Region: Cambodia, Laos, Thailand, Vietnam.
Palaearctic Region: China (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
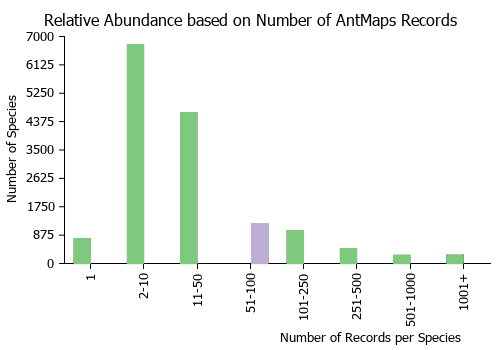
|
Biology
Castes
Known only from the worker caste.
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- fuchuanensis. Aenictus fuchuanensis Zhou, 2001b: 59, 231, figs. 74, 75 (w.) CHINA (Guangxi).
- Type-material: holotype worker, 19 paratype workers.
- Type-locality: holotype China: Guangxi, Fuchuan County, 18.vi.1998 (S. Zhou); paratypes with same data.
- Type-depository: GNUC.
- [Misspelled as fuhuanensis by Jaitrong, et al. 2011: 321.]
- Status as species: Wang, W. 2006: 637 (in key); Guénard & Dunn, 2012: 22; Jaitrong & Yamane, 2013: 187 (redescription); Staab, 2015: 141 (in key); Jaitrong, Guénard, et al. 2016: 24.
- Distribution: Cambodia, China, Laos, Thailand.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Holotype: TL 3.5, HL 0.66, HW 0.72, CI 109, SL 0.56, SI 77, PW 0.40, AL 1.10. Paratypes: TL 3.2~3.5, HL 0.62~0.66, HW 0.68~0.72, CI 104~109, SL 0.53~0.56, SI 77~80, PW 0.36~0.40, AL 1.08~1.10.
Head slightly broader than long, sides and occipital border convex. Mandible narrow, with a large apical tooth and 3 small teeth. There remains a gap between posterior borders of mandibles and center of anterior clypeal border 2 X as wide as maximum width of mandible. Clypeus narrow, anterior border straight, without any tooth. Frontal lobes narrowly apart. Antennal scapes nearly reaching occipital border of head. Pronotum convex above, promesonotal suture vestigial, mesopropodeal suture incised; basal face of propodeum straight, declivity concave, conjunction between them acutely angular, with low transverse ridge. Petiolar node convex dorsally, elevate posteriorly; Subpetiolar process low and indistinct. Gaster longitudinally oval. Sting developed. Mandibles with fine longitudinal striations; head smooth and shining; alitrunk, petiole and post petiole with coarse reticulations; mesopleura and dorsal of propodeum with several longitudinal striations; gaster smooth and shining. Erect and suberect hairs abundant.
Color reddish brown, alitrunk, petiole and postpetiole darker.
Jaitrong and Yamane (2013) - (n = 10). TL 3.10–3.45 mm; HL 0.63–0.70 mm; HW 0.70–0.75 mm; SL 0.53–0.58 mm; ML 0.98–1.05 mm; PL 0.25–0.28 mm CI 107– 112; SI 75–79.
Head in full-face view slightly shorter than broad, anterior portion slightly broader than posterior portion, sides convex and posterior margin almost straight; occipital margin bearing a carina. Antennal scape relatively long, reaching 2/3 of head length. Frontal carina short, slightly extending beyond the level of posterior margin of torulus. Masticatory margin of mandible with 4 teeth, including a large apical tooth; basal margin feebly concave. Maximum width of gap between anterior clypeal margin and mandibles about 2 times as broad as maximum width of mandible. Promesonotum strongly convex dorsally and sloping gradually to metanotal groove that is indistinct; metapleural gland bulla relatively small, its maximum diameter about 2 times as long as distance between propodeal spiracle and metapleural gland bulla. Propodeum in profile with almost straight dorsal outline; propodeal junction acutely angulated; declivity of propodeum broad, shallowly concave, and encircled with a distinct rim. Petiole cylindrical, distinctly longer than high, with its dorsal outline slightly elevated posteriorly, posterior face of petiole flat, and encircled with a indistinct carina; subpetiolar process generally weakly developed, seen in profile its ventral outline almost straight or weakly convex, and anteroventral corner acutely angulated. Postpetiole almost as long as petiole, with its dorsal outline elevated posteriorly.
Head entirely smooth and shiny. Mandible with fine longitudinal striation. Basal half of antennal scape microreticulate; apical half smooth and shiny. Promesonotum reticulate except anteriormost portion punctate; mesopleuron, metapleuron and propodeum punctuate; in addition mesopleuron and metapleuron with approximately 10 longitudinal rugae. Petiole and postpetiole densely punctate. Femora entirely smooth and shiny except punctate basal portion; tibiae entirely smooth and shiny.
Head and mesosoma dorsally with relatively dense standing hairs; longest pronotal hair 0.23–0.25 mm long. Head and mesosoma dark reddish brown; petiole, postpetiole and antenna reddish brown; gaster and legs yellowish brown, paler than head, mesosoma and waist; mandible dark brown.
Type Material
Jaitrong and Yamane (2013) - CHINA: Holotype and 19 paratype workers from Fuchaun County, Guangxi, 18.VI.1998, Shanyi Zhou leg (not examined).
References
- Borowiec, M.L. 2019. Convergent evolution of the army ant syndrome and congruence in big-data phylogenetics. Systematic Biology 68, 642–656 (doi:10.1093/sysbio/syy088).
- Jaitrong, W.; Yamane, S. 2013. The Aenictus ceylonicus species group (Hymenoptera, Formicidae, Aenictinae) from Southeast Asia. Journal of Hymenoptera Research 31:165-233.
- Khachonpisitsak, S., Yamane, S., Sriwichai, P., Jaitrong, W. 2020. An updated checklist of the ants of Thailand (Hymenoptera, Formicidae). ZooKeys 998, 1–182 (doi:10.3897/zookeys.998.54902).
- Jaitrong, W. & Yamane, S. 2011. Synopsis of Aenictus species groups and revision of the A. currax and A. laeviceps groups in the eastern Oriental, Indo-Australian, and Australasian regions (Hymenoptera: Formicidae: Aenictinae). Zootaxa, 3128, 1–46. PDF
- Zhou, S. 2001. Ants of Guangxi. Guilin, China: Guangxi Normal University Press. 255 pp. (page 59, 231, figs. 74, 75 worker described)
References based on Global Ant Biodiversity Informatics
- Guénard B., and R. R. Dunn. 2012. A checklist of the ants of China. Zootaxa 3558: 1-77.
- Jaitrong W. 2015. A revision of the Thai species of the ant genus Aenictus Shuckard, 1840 (Hymenoptera: Formicidae: Dorylinae). The Thailand Natural History Museum Journal 9(1): 1-94.
- Jaitrong W., B. Guenard, E. P. Economo, N. Buddhakala, and S. Yamane. 2016. A checklist of known ant species of Laos (Hymenoptera: Formicidae). Asian Myrmecology 8: 1-32. DOI: 10.20362/am.008019
- Jaitrong W., and S. Yamane. 2013. The Aenictus ceylonicus species group (Hymenoptera, Formicidae, Aenictinae) from Southeast Asia. Journal of Hymenoptera Research 31: 165-233.
- Skarbek C. J., M. Noack, H. Bruelheide, W. Hardtle, G. von Oheimb, T. Scholten, S. Seitz, M. Staab. 2019. A tale of scale: plot but not neighbourhood tree diversity increases leaf litter ant diversity. Journal of Animal Ecology DOI: 10.1111/1365-2656.13115
- Staab M. 2015. Aenictus hoelldobleri sp. n., a new species of the Aenictus ceylonicus group (Hymenoptera, Formicidae) from China, with a key to the Chinese members of the group. Zookeys 516: 137-155.
- Zhang R. J., L. W. Liang, and S. Y. Zhou. 2014. An analysis on the ant fauna of Nonggang Nature Reserve in Guangxi, China. Journal of Guangxi Normal university: Natural Science Edition 32(3): 86-93.
- Zhou S.-Y. 2001. Ants of Guangxi. Guangxi Normal University Press, Guilin, China, Guilin, China. 255 pp.