Amoimyrmex silvestrii
| Amoimyrmex silvestrii | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Attini |
| Genus: | Amoimyrmex |
| Species: | A. silvestrii |
| Binomial name | |
| Amoimyrmex silvestrii (Emery, 1905) | |
| Synonyms | |
| |
Found in open and dry habitats of the Chaco and Pampas. The nest entrance is a simple hole in the soil surface, never with a mound or crater, with all vegetation removed from around the perimeter.
Identification
Cristiano, Cardoso and Sandoval (2020) - Workers can be distinguished from congeners by combination of the following features: body reddish‐brown to dark brown with dark brown gaster; pronotum without median pronotal projections, best seen in frontal view; shiny integument covered by rough irregular striae and sparse yellowish non‐decumbent setae, varying in length.
Am. silvestrii can easily be distinguished from the sympatric Amoimyrmex bruchi by its larger size, which is more evident in major workers, absence of median pronotal spines (always present and well developed in Ac. bruchi), head with rounded posterior cephalic corners (laterally sub‐rectangular in bruchi and striatus), less non‐decumbent pilosity, mainly on the metasoma, compared to Am. bruchi, and irregular striae restricted to the antero‐central portion of the first gastral tergite (more broadly distributed in Am. bruchi).
Keys including this Species
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: -16° to -37.12°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: Argentina (type locality), Uruguay.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
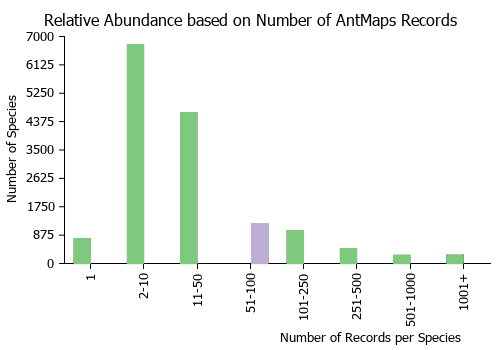
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
|
Biology
|
Cristiano, Cardoso and Sandoval (2020) - Occurs in sympatry with Am. bruchi (Brandão 1991; Delabie et al. 2011; Simões‐Gomes et al. 2017), in the Chaco and Pampas and always nests in open and dry habitats. External appearance of nests similar to those of its congeners, never with mounds or craters. The entrances are simple holes in the soil surface with all vegetation removed from around their perimeters. Nest architecture was previously described by Weber as several subterranean chambers connected by tunnels. The fungus gardens are restricted to the deeper chambers, where the fungus is suspended from the ceilings. None of the superficial chambers contain fungus, only workers and brood. Armani and Quirán (2007) suggested that Am. silvestrii (as Ac. striatus) is a generalist species, cutting small quantities of available monocots.
Barrera et al. (2015) studied the diversity of leaf cutting ants along a forest-edge-agriculture habitat gradient. Their study site, in Chaco Serrano of Central Argentina, had forest remnants of various sizes within an agriculture area with wheat, soy and maize. A few colonies of A. silvestrii and Acromyrmex heyeri were found along the forest edge. Acromyrmex crassispinus had the highest occurance (42% of 162 colonies) and was most abundant in the forest interior. Acromyrmex lundii and A. striatus were also present, with the former also found in the forest interior. The latter and Amoimyrmex striatus were the most abundant in the forest edge habitat with A. crassispinus present at a slightly lower density.
Castes
Worker
Reproductive Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- silvestrii. Atta (Moellerius) silvestrii Emery, 1905c: 42 (w.) ARGENTINA (Córdoba).
- [Also noted as new by Emery, 1906c: 166. Nomen nudum.]
- [Misspelled as sylvestrii by Cherrett & Cherrett, 1989: 52.]
- Forel, 1911c: 292 (q.m.); Santschi 1912e: 530 (q.m.).
- Combination in Acromyrmex (Moellerius): Forel, 1913l: 236.
- combination in Amoimyrmex: Cristiano, Cardoso & Sandoval, 2020: 662.
- Subspecies of striatus: Gallardo, 1916d: 338; Santschi, 1920d: 380.
- Status as species: Forel, 1911c: 292; Forel, 1912e: 180; Santschi, 1912e: 530; Forel, 1913l: 236; Bruch, 1914: 217; Bruch, 1915: 529; Bruch, 1916: 326; Santschi, 1916e: 389; Bruch, 1923: 198; Emery, 1924d: 351; Santschi, 1925a: 389 (in key); Kempf, 1972a: 16; Zolessi, et al. 1988: 5; Fowler, 1988: 290; Cherrett & Cherrett, 1989: 52; Brandão, 1991: 323; Bolton, 1995b: 57.
- Senior synonym of mesopotamicus: Fowler, 1988: 290; Brandão, 1991: 323; Bolton, 1995b: 57.
- mesopotamicus. Acromyrmex (Moellerius) mesopotamicus Gallardo, 1916d: 337, fig. 3 (w.) ARGENTINA (Entre Ríos).
- Status as species: Emery, 1924d: 351; Santschi, 1925a: 389 (in key); Kempf, 1972a: 16; Cherrett & Cherrett, 1989: 51 (error).
- Junior synonym of silvestrii: Fowler, 1988: 290; Brandão, 1991: 323; Bolton, 1995b: 56.
Type Material
Cristiano, Cardoso and Sandoval (2020) - We designate the following syntype (worker) as the lectotype 1☿(Museo Civico di Storia Naturale, Genoa) ‘La Carlota, silvestrii/Atta silvestrii Emery/ANTWEB CASENT0905007’ [image examined]. Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Cristiano, Cardoso and Sandoval (2020) - Range (specimen used for redescription): TL 4.02–8.71 (8.06), HL 0.82–2.00 (1.76), HW 0.92–2.29 (2.09), ML 0.35–0.85 (0.68), SL 0.82–1.82 (1.68), EL 0.16– 0.35 (0.29), WL 1.20–2.85 (2.47), PL 0.26–0.74 (0.68), PW 0.25–0.62 (0.50), PPL 0.22–0.63 (0.56), PPW 0.45–1.00 (0.91), GL 0.98–2.21 (1.91), CI 100.00–133.33 (118.33), MI 31.67–46.77 (38.33), OI 12.66–19.61 (14.08), SI 88.00– 117.65 (95.00) [N = 82].
Head. In full‐face view, posterior cephalic margin medially emarginate. Posterior cephalic corner rounded, with a small and sharp spine directed upwards, rising on the top of head in lateral view, and a small tubercle laterad. Integument covered by rough irregular striae and sparse yellowish non‐decumbent setae, varying in length. Frontal carina, that may be confused with striae, extending to vertex and almost reaching posterior cephalic corner. Mandible with 8–11 teeth (some can be considered denticles, varying in size and position), dorsally glossy, striated and covered by light‐coloured hairs, with larger ones at the masticatory margin. Eye convex, 16–24 ommatidia across largest diameter. Frontal lobe partially covering antennal insertions, with a hook shape. Antennal scrobe absent. Antennal scape slightly surpassing the posterior cephalic margin, less than 1/3 of its length (SI = 88.00–117.65). Colour reddish‐brown. Mesosoma. Pronotum armed with two large lateral pronotal spines and without median pronotal projections, best seen in frontal view. Mesonotum with two lateral mesonotal spines and two posterior mesonotal spines, with similar size and less than half of size of lateral pronotal spines. Propodeum bearing two large sharp spines, moderately larger than lateral pronotal spines. Integument covered by rough irregular striae and sparse non‐decumbent pilosity, reaching up the spines. Legs with conspicuous reticulation, in some cases with coxa and femur darker than the rest of body. Metasoma. Petiolar node with four spine‐like projections directed up and backwards and two lateral longitudinal carinae. Postpetiole node with two small teeth on lateral margins, located in medial portion, projected to the sides and slightly backwards, best seen in dorsal view, and two lateral longitudinal carinae. Dorso‐posterior region of node with up to four small tuberculiform projections. Integument of petiole and postpetiole covered by irregular striae and non‐decumbent pilosity. Gaster darker than rest of body with smooth and shiny integument and dense yellowish non‐decumbent setae of two sizes, the longest ones twice the length of short ones. Antero‐central portion of first gastral tergite with irregular striae, discernible in medium magnification (~50×), bearing sometimes a pair of reddish to yellowish spots laterad.
Queen
Cristiano, Cardoso and Sandoval (2020) - TL 9.50–10.70, HL 1.75–1.95, HW 2.25–2.30, ML 0.80–1.00, SL 1.50–1.65, EL 0.35–0.40, WL 2.65–3.25, PL 0.73–0.85, PW 0.65–0.75, PPL 0.65–0.78, PPW 1.05–1.30, GL 2.70–3.10, CI 115.38–131.43, MI 43.24– 54.05, OI 15.56–17.78, SI 81.08–84.61 [N = 10].
Head. In full‐face view, posterior cephalic margin medially emarginate but less accentuated than in worker. Posterior cephalic corner rounded, with a small spine directed upwards and an inconspicuous tubercle directed laterad. Integument covered by rough irregular striae, as in worker. Frontal carina present but shorter than in worker. Eye larger than in worker. Number of ommatidia across largest eye diameter 24– 28. Mandibles with 10 teeth (two can be considered denticles) and very large and sharp apical tooth, four times more prominent than in worker but less striate. Mesosoma. Integument with well‐defined longitudinal irregularstriae, denser and more parallel than in worker, and sparse non‐decumbent pilosity. Scutum in dorsal view with reduced notauli forming a shallow impression. Median mesoscutal line with only anterior portion visible as a slightmark on the integument. Scutellum strongly convex in lateral view, narrowing posteriorly with a trapezoidal outline in dorsal view, bearing two tubercles in the posterior margin. Propodeum with a pair of protruding long spines, directed upwards, in lateral view. Metasoma. Petiole with a well‐developed subpetiolar process armed with a truncated spine. Integument of postpetiole covered by transversestriae. First gastral tergite with two yellowish spots located anterolaterally in dorsal view. Presence of semicircular striae on the first gastral tergite, mainly at the base. Dense yellowish non‐decumbentsetae of two sizesin all gastral tergites, being denser in the apical ones.
Male
Cristiano, Cardoso and Sandoval (2020) - TL 7.80–7.99, HL 1.13–1.16, HW 1.33–1.43, ML 0.50–0.52, SL 1.28–1.31, EL 0.38–0.40, WL 2.50–2.53, PL 0.60–0.62, PW 0.73–0.77, PPL 0.33–0.42, PPW 1.13–1.17, GL 2.70–2.74, CI 122.22–123.27, MI 44.44–44.82, OI 27.27–27.97, SI 112.93–113.33 [N = 8].
Head. In full‐face view subquadrate, posterior cephalic margin straight, without emargination. Posterior cephalic corner rounded with a small spine directed upwards. Integument opaque, reticulated and covered by irregularstriae, denser than in worker and queen. Eye bigger and more convex than worker and queen. Number of ommatidia across largest eye diameter 34–35. Mandibles elongate with two well‐developed teeth at apex and some sparse denticles along the internal margin. Mesosoma. Scutellum convex, less so than in queen in lateral view, narrowing posteriorly with a trapezoidal outline in dorsal view, bearing two tuberculiform denticles on posterior margin. Propodeum with a pair of protruding long spines, directed backwards, in lateral view. Integument with well‐defined longitudinal and parallel striae, non‐decumbent pilosity in scutum. Fine decumbent pilosity scarce on pronotum and denser at the base of propodeal spines. Metasoma. Petiole with short peduncle, subpetiolar process smaller than in queen and not forming a spine. Postpetiolar process well developed, larger than in queen. Integument of petiole and postpetiole with scarce irregular striae. Gaster with smooth and shiny integument, without tubercles and striae. Non‐decumbent pilosity less dense and shorter than in queen.
Karyotype
- 2n = 22, karyotype = 20M+2SM (Argentina) (Micolino et al., 2021).
References
- Barrera, C. A., L. M. Buffa, and G. Valladares. 2015. Do leaf-cutting ants benefit from forest fragmentation? Insights from community and species-specific responses in a fragmented dry forest. Insect Conservation and Diversity. 8:456-463. doi:10.1111/icad.12125
- Cardoso, D. C., Cristiano, M. P. 2021. Karyotype diversity, mode, and tempo of the chromosomal evolution of Attina (Formicidae: Myrmicinae: Attini): Is there an upper limit to chromosome number? Insects 1212, 1084 (doi:10.3390/insects12121084).
- Cristiano, M.P., Cardoso, D.C., Sandoval‐Gómez, V.E., Simões‐Gomes, F.C. 2020. Amoimyrmex Cristiano, Cardoso, Sandoval, gen. nov. (Hymenoptera: Formicidae): a new genus of leaf‐cutting ants revealed by multilocus molecular phylogenetic and morphological analyses. Austral Entomology 59, 643–676 (doi:10.1111/aen.12493).
- Emery, C. 1905f. Revisione delle specie del genere Atta appartenenti ai sottogeneri Moellerius e Acromyrmex. Mem. R. Accad. Sci. Ist. Bologna (6)2:39-54 (page 42, worker described)
- Emery, C. 1924f [1922]. Hymenoptera. Fam. Formicidae. Subfam. Myrmicinae. [concl.]. Genera Insectorum 174C: 207-397 (page 351, Revived status as species)
- Forel, A. 1911e. Ameisen des Herrn Prof. v. Ihering aus Brasilien (Sao Paulo usw.) nebst einigen anderen aus Südamerika und Afrika (Hym.). Dtsch. Entomol. Z. 1911: 285-312 (page 292, queen, male described)
- Forel, A. 1913m. Fourmis d'Argentine, du Brésil, du Guatémala & de Cuba reçues de M. M. Bruch, Prof. v. Ihering, Mlle Baez, M. Peper et M. Rovereto. Bull. Soc. Vaudoise Sci. Nat. 49: 203-250 (page 236, Combination in Acromyrmex (Moellerius))
- Forti, L.C., Andrade, A.P.P., da Silva Camargo, R., Filho, T.M.M.M., Scudillio, T.T., Zanuncio, J.C., Sousa, K.K.A., Caldato, N. 2022. Taxonomic revision of the leaf-cutting ant Acromyrmex subterraneus (Forel, 1893). International Journal of Tropical Insect Science (doi:10.1007/s42690-022-00815-5).
- Fowler, H. G. 1988b. Taxa of the neotropical grass-cutting ants, Acromyrmex (Hymenoptera: Formicidae: Attini). Científica (Jaboticabal) 16: 281-295 (page 290, senior synonym of mesopotamicus)
- Micolino, R., Baldez, B.C.L., Sánchez-Restrepo, A.F., Calcaterra, L., Cristiano, M.P., Cardoso, D.C. 2021. Karyotype structure and cytogenetic markers of Amoimyrmex bruchi and Amoimyrmex silvestrii: contribution to understanding leaf-cutting ant relationships. Genome 651, 43–51 (doi:10.1139/gen-2021-0044).
- Mueller, U.G., Ishak, H.D., Bruschi, S.M., Smith, C.C., Herman, J.J., Solomon, S.E., Mikheyev, A.S., Rabeling, C., Scott, J.J., Cooper, M., Rodrigues, A., Ortiz, A., Brandão, C.R.F., Lattke, J.E., Pagnocca, F.C., Rehner, S.A., Schultz, T.R., Vasconcelos, H.L., Adams, R.M.M., Bollazzi, M., Clark, R.M., Himler, A.G., LaPolla, J.S., Leal, I.R., Johnson, R.A., Roces, F., Sosa-Calvo, J., Wirth, R., Bacci, M. 2017. Biogeography of mutualistic fungi cultivated by leafcutter ants. Molecular Ecology 26, 6921–6937 (doi:10.1111/mec.14431).
- Santschi, F. 1912e. Quelques fourmis de l'Amérique australe. Rev. Suisse Zool. 20: 519-534 (page 530, queen, male described)
- Santschi, F. 1920d [1919]. Formicides africains et américains nouveaux. Ann. Soc. Entomol. Fr. 88: 361-390 (page 380, Stirps of striatus)
References based on Global Ant Biodiversity Informatics
- Bruch C. 1914. Catálogo sistemático de los formícidos argentinos. Revista del Museo de La Plata 19: 211-234.
- Bruch C. 1915. Suplemento al catálogo de los formícidos argentinos. I. (Addenda et corrigenda). Revista del Museo de La Plata 19: 527-537.
- Bruch C. 1916. Contribución al estudio de las hormigas de la provincia de San Luis. Revista del Museo de La Plata 23: 291-357.
- Cuezzo, F. 1998. Formicidae. Chapter 42 in Morrone J.J., and S. Coscaron (dirs) Biodiversidad de artropodos argentinos: una perspectiva biotaxonomica Ediciones Sur, La Plata. Pages 452-462.
- Culebra Mason S., C. Sgarbi, J. Chila Covachina, J. M. Pena, N. Dubrovsky Berensztein, C. Margaria, and M. Ricci. 2017. Acromyrmex Mayr (Hymenoptera: Formicidae: Myrmicinae): species distribution patterns in the province of Buenos Aires, Argentina. Rev. Mus. Argentino Cienc. Nat. 19(2) 185-199.
- Fernández, F. and S. Sendoya. 2004. Lista de las hormigas neotropicales. Biota Colombiana Volume 5, Number 1.
- Forel A. 1912. Formicides néotropiques. Part II. 3me sous-famille Myrmicinae Lep. (Attini, Dacetii, Cryptocerini). Mémoires de la Société Entomologique de Belgique. 19: 179-209.
- Forel A. 1913. Fourmis d'Argentine, du Brésil, du Guatémala & de Cuba reçues de M. M. Bruch, Prof. v. Ihering, Mlle Baez, M. Peper et M. Rovereto. Bulletin de la Société Vaudoise des Sciences Naturelles. 49: 203-250.
- Gallardo A. 1916. Notes systématiques et éthologiques sur les fourmis attines de la République Argentine. Anales del Museo Nacional de Historia Natural de Buenos Aires 28: 317-344.
- Kempf, W.W. 1972. Catalago abreviado das formigas da regiao Neotropical (Hym. Formicidae) Studia Entomologica 15(1-4).
- Marcus H. 1945. La diferencia en la fauna de las hormigas de Cochabamba y Liriuni. Revista de Agricultura 3: 28-29.
- Santschi F. 1912. Quelques fourmis de l'Amérique australe. Revue Suisse de Zoologie 20: 519-534.
- Santschi F. 1925. Revision du genre Acromyrmex Mayr. Revue Suisse de Zoologie 31: 355-398.
- Vittar, F. 2008. Hormigas (Hymenoptera: Formicidae) de la Mesopotamia Argentina. INSUGEO Miscelania 17(2):447-466
- Vittar, F., and F. Cuezzo. "Hormigas (Hymenoptera: Formicidae) de la provincia de Santa Fe, Argentina." Revista de la Sociedad Entomológica Argentina (versión On-line ISSN 1851-7471) 67, no. 1-2 (2008).
- Zolessi L. C. de, Y. P. Abenante, and M. E. de Philippi. 1988. Lista sistematica de las especies de Formicidos del Uruguay. Comun. Zool. Mus. Hist. Nat. Montev. 11: 1-9.
- Zolessi L. C. de; Y. P. de Abenante, and M. E. Philippi. 1989. Catálogo sistemático de las especies de Formícidos del Uruguay (Hymenoptera: Formicidae). Montevideo: ORCYT Unesco, 40 + ix pp.
- de Zolessi, L.C., Y.P. de Abenante and M.E. Philippi. 1987. Lista sistemática de las especies de formícidos del Uruguay. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo 11(165):1-9





