Dinoponera lucida
| Dinoponera lucida | |
|---|---|

| |
| Conservation status | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Ponerinae |
| Tribe: | Ponerini |
| Genus: | Dinoponera |
| Species: | D. lucida |
| Binomial name | |
| Dinoponera lucida Emery, 1901 | |
Dinoponera lucida has been classified as endangered in Brazil by the Ministério do Meio Ambiente (Campiolo et al. 2003) due to habitat destruction in the Atlantic forest.
Identification
Lenhart et al. (2013) - Worker. This species can be recognized by the following combination of character states: anterior inferior pronotal corner with tooth-like process, pilosity long and flagellate with white luster, integument smooth and shiny with bluish luster, scape length longer than head width, petiole slanting obliquely on dorsal edge. Total body length ranges from 27–30mm which is between the lengths of Dinoponera australis and the other larger species.
Dinoponera lucida is only slightly larger than D. australis but differs in its integument micro-sculpturing and pilosity type (see above). Dinoponera lucida can be confused with D. australis but is distinguished by its shiny integument and whitish setae, as opposed to the micro-sculptured integument and dull tan setae of D. australis.
Key to Dinoponera workers / Clave para la identificación de las obreras de Dinoponera / Chave para identificação de operários de Dinoponera
Escarraga et al. (2017) - Male Body bicolored: head, mesosoma, and petiole brown; scape, legs and gaster ferruginous brown. Lateral ocelli protruding beyond posterior cephalic margin in dorsal view of the head; abdominal tergum VIII shaped as acute triangular; body with abundant long yellow hairs.
Keys including this Species
Distribution
Escarraga et al. (2017) - Endemic to the Atlantic Forest of Brazil, recorded from the states of Bahia, Espírito Santo, Minas Gerais, and São Paulo, with most records in Espírito Santo and Southern Bahia. The most northern record is from Ilheus (Bahia) and the southernmost record is from Cruzeiro (São Paulo). There is a single record from São Paulo by Kempf (1971).
Lenhart et al. (2013) - This species inhabits fragments of Atlantic rainforest in the Brazilian state of Espirito Santo, across the border into Minas Gerais, the southern portion of Bahia and São Paulo. It is possible that D. lucida exists in Rio de Janeiro but we are not aware of any specimens from this area. Refer to Mariano et al. (2008) for information on the biogeography of this species. With the locality data available D. lucida is the only species with no known range overlaps with other Dinoponera species.
Latitudinal Distribution Pattern
Latitudinal Range: -16.13333333° to -20.29961623°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: Brazil (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
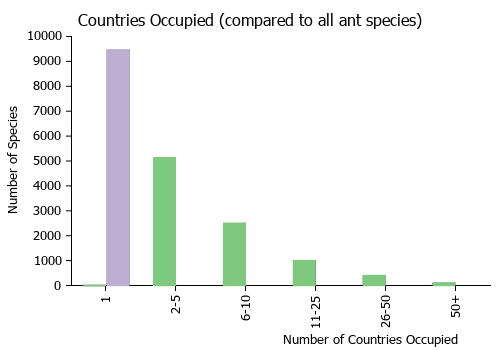
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
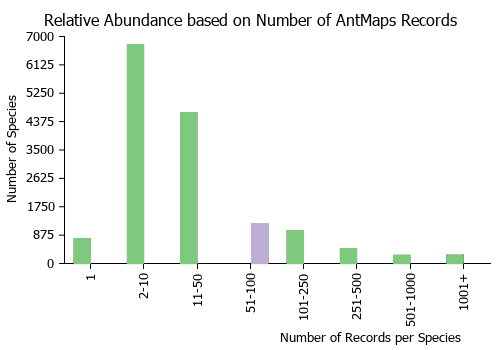
|
Biology
Dinoponera lucida is an Atlantic Forest endemic, with most records from the state of Espírito Santo, that has received official recognition as an endangered species (Campiolo & Delabie 2008) due to its restricted distribution within an ecosystem that is dwindling in size and suffering from fragmentation (Saatchi et al. 2001; Ribeiro et al. 2009). Several aspects of the species biology have been studied by Peixoto et al. (2008) and Peixoto et al. (2010). Amongst their results, they found a preference of these ants for dense forests with little herbaceous ground vegetation. Mature nests have sizes that range from 22 to 106 adults, with a nest density of 20–52 nests per hectare.
Associations with other Insects
Scuttle flies (Diptera: Phoridae) have been found attacking workers of this ant species (Disney et al. 2015). Marcos Teixeira collected Diptera of Apocephalus exlucida and two undescribed species of the genus Megaselia attacking and hovering over Dinoponera lucida in the vicinity of study colonies at Aracruz, Espírito Santo State, in David Farina Park 19°55′54″S 40°07′41″W / 19.93167°S 40.12806°W.
- This species is a host for the eucharitid wasp Kapala sp. (a parasite) (Universal Chalcidoidea Database) (primary host).
- This species is a host for the phorid fly Apocephalus exlucida (a parasitoid) (Quevillon, 2018) (encounter mode primary; direct transmission; transmission outside nest).
- This species is a host for the phorid fly Megaselia sp. A (a parasitoid) (Quevillon, 2018) (encounter mode primary; direct transmission; transmission outside nest).
- This species is a host for the phorid fly Megaselia sp. B (a parasitoid) (Quevillon, 2018) (encounter mode primary; direct transmission; transmission outside nest).
Castes
Male

| |
| . | |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- lucida. Dinoponera grandis subsp. lucida Emery, 1901a: 48 (w.) BRAZIL (Espirito Santo).
- Type-material: syntype workers (number not stated).
- Type-locality: Brazil: Espirito Santo (ex coll. Fruhstorfer).
- [Note: Kempf, 1971: 376, proposes the type-locality as, “Cidade do Espirito Santo, better known under the name of Vila Velha, near Vitória, Espirito Santo State”.]
- Type-depository: MSNG.
- Escárraga, et al. 2017: 130 (m.).
- Subspecies of grandis: Forel, 1907e: 1; Emery, 1911d: 63; Mann, 1916: 408; Santschi, 1921g: 84; Borgmeier, 1923: 63.
- Subspecies of gigantea: Borgmeier, 1937b: 226.
- Status as species: Kempf, 1971: 376 (redescription); Kempf, 1972a: 97; Bolton, 1995b: 171; Lenhart, et al. 2013: 146 (redescription); Feitosa, 2015c: 98; Escárraga, et al. 2017: 130; Dias, A.M. & Lattke, 2021: 38 (redescription).
- Distribution: Brazil.
Type Material
Lenhart et al. (2013) - Syntype workers BRASIL: Espírito Santo, ex coll Fruhstorfer. (Museo Civico di Storia Naturale, Genoa) [examined]. (specific locality of Vila Velha (Cidade do Espirito Santo) proposed by Kempf (1971). Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
(mm) (n=5) TBL: 27.01–30.39 (28.64); MDL: 3.79–4.31 (3.97); HL: 4.92–5.64 (5.34); HW: 5.02–5.13 (5.07); SL: 5.23–5.64 (5.42); WL: 7.33–8.20 (7.84); PL: 2.25–2.51 (2.39); PH: 3.18–3.28 (3.26); PW: 1.54–1.90 (1.72); GL: 8.00–10.05 (9.10); HFL: 6.87–7.28 (7.18).
A description of the external morphology of the worker is given in Kempf (1971): “Antennal scape distinctly longer than head width. Pubescence on front and vertex of head variable, either short and inconspicuous or longer, denser and quite visible. Gular surface of head finely striate either throughout or at least on anterior half; very seldom the striae are confined to a narrow stripe along the anterior border and obsolescent yet still discernible. Sides of head smooth, not quite glossy but with a silky sheen on account of the superficial reticulate microsculputure. Antero-inferior corner of pronotum dentate. Pronotal disc smooth and shining, lacking wrinkles and dense, fine punctulae; paired swellings quite distinct. Hind tarsus I decidedly longer than head length. Petiole…smooth and polished, its anterior face not excavate, its dorsal face slanted forward; vertical sulcus on posterior face either present or absent; width-length proportion well under 0.80; anterior face lacking dense pubescence. Terga I and II of gaster smooth, highly shining, lacking dense, fine punctulae; pubescence loosely scattered on sides, entirely absent on disc. Stridulatory file on acrotergite of tergum II well developed, nearly crossing the entire tergite.”
Male
Escarraga et al. (2017) - (n=8) HL= 1.90–2.10, HW1= 1.58–1.86, HW2= 2.22–2.44, MAL= 0.14–0.24, MDL= 0.48–0.56, SL= 0.60–0.76, PDL= 0.20–0.24, A3L= 1.38–1.50, A4L= 1.40–1.58, AAL= 1.34–1.46, EL= 1.16–1.28, EW= 0.74–0.80, MOD= 0.34–0.40, OOD= 0.32–0.42, LOD= 0.32–0.38, WL= 5.83–6.33, MLL= 1.14–1.48, MLW= 2.00–2.26, MTL= 2.52–2.68, MTW= 2.56–2.84, PFL= 3.68–3.96, MFL= 4.72–5.08, FWL= 13.3–15.0, HWL= 10.0–11.7, PTH= 1.24–1.48, PTL= 1.66–1.82, PTW= 0.98–1.10, GL= 7.33–8.17, TBL= 17.6–18.7, CI=74.7–93.9, CS= 1.66–1.95, SEI= 168–200, SI= 32.3–46.5, EI1= 59.7–63.8, EI2= 98–119, MDI= 23.1–28.4, OBI=60.2–79.3, OMI= 483–900, MNI= 1.49–1.84, MTI= 101–184 FI= 76.7–80.5, PTI= 70–87.1.
Cephalic capsule ovoid in dorsal view, wider than long including compound eyes; posterior cephalic margin forming blunt angle interrupted by lateral ocelli close to its apex; lateral head margin convex, formed by compound eye. Clypeus protrudes posteriorly between compound eyes; anterior margin bordered by narrow, translucent lamella; lamella medially straight to weakly convex, laterally convex; surface of clypeal disc very convex, epistomal sulcus posteriorly convex and weakly impressed, separating clypeus from weakly elevated but flattened frontal triangle that extends posterad between antennal sockets; anterior tentorial pit large and easily discernible. Frontal carina reduced to faint and short elevated line, median torular arch shaped as weak posteromedian convex lobe that leaves antennal condyle completely exposed, lateral torular arch simple; antennal socket located at cephalic mid-length, as measured from anterior clypeal margin to posterior head margin, separated from other socket by distance shorter than median ocellus diameter (MOD= 0.34–0.40). Compound eye large and bulging (EI2= 98–119), occupying whole lateral cephalic margin (OBI= 60.2–79.3), dorsomedial margin weakly emarginate; ocelli large and protruding, approximately equal in sized, lateral ocelli closer to anterior ocellus than to each other and separated from compound eye by a distance similar to its diameter (OOD=0.32–0.42). Antenna longer than mesosoma, 13-segmented; scape length approximately half of first flagellomere and shorter than EL; pedicel length approximately one-third that of scape, surface smooth; flagellum filiform, punctate. Palpal formula 4,3 (in situ count); labrum, in dorsal view, lateromedially narrower than labium, weakly projecting as transverse, anteriorly emarginate structure with convex dorsal surface. Mandible slightly shorter than antennal scape, apically rounded, lacking teeth, surface smooth and shiny, with basal, flattened lateral expansion. Cephalic sculpture mostly shining with scattered small, piligerous tubercles; weak longitudinal striae present between ocelli and antenna.
Mesosoma in lateral view with anterior pronotal margin forming brief overhang above head, narrow posterior shelf present at promesonotal junction; mesoscutum convex; mesoscutellum strongly convex; metanotum brief and strongly convex; propodeal dorsal margin straight to weakly convex in profile view, forming smooth continuity with broadly convex declivitous margin.
Pronotum trapezoid in lateral view, posterolateral corners blunt and slighted lifted; mesopleural sulcus oblique, deeply impressed, scrobiculate, mesopleural pit directed ventrally; spiracular sclerite oval, directed anterolaterally; metapleuron medially constricted, with conspicuous and deep pit medially located near metapleural-propodeal suture; metapleural-propodeal suture broad and shallow but well-defined; propodeal spiracle slit-shaped. In dorsal view, pronotal posterior margin broadly concave, lateral lobe protruding, mesoscutum with three longitudinal carinae: one anterior median notal carina and two lateral parapsidal lines; notauli absent, mesoscutum smooth and shining; scutoscutellar sulcus scrobiculate, mesoscutellum hexagonal, disc smooth and shining; metascutellum transversally rectangular with transverse striae. Mesosoma mostly smooth and shining except for metanotum and irregular minute raised areas on propodeal declivity. Protibial calcar pectinate with small basal lamella (= “velum of calcar”), probasitarsus with basal comb; meso- and metatibia each with long pectinate spur and simple short spur; all basitarsi long, each about same length of its corresponding tibia; tarsal claws with preapical tooth; arolium well-developed.
Fore wing with pterostigma, abscissae C, Sc+R, R, Rs, Rs+M, M+Cu, M, Cu, and A present; crossveins 2r-rs, 2rs-m, 1m-cu, cu-a present. Cells Costal+Subcostal, Basal, Subbasal, Marginal, Submarginal 1, Submarginal 2, Discal, Subdiscal present. Rs weakly curved anterad R; M and Cu curved posterad, without reaching posterior margin. Crossvein cu-a far from wing base, near discal cell (Fig. 3A). Hind wing with abscissae R+Rs, Rs, M+Cu, M, Cu, and A present; crossveins 1rs-m and cu-a present; no abscissae reach posterior margin; segment of M+Cu
posterior to cu-a twice as long as segment of M anterior to rs-m; 1rs-m almost twice longer than cu-a, jugal lobe present (Fig. 3B).
Petiole rounded in lateral view, anterior margin more broadly convex than posterior margin, petiolar spiracle relatively small, oval, anteriorly located. Petiole rectangular in dorsal view, longer than broad with anterior cervix developed, posterior corner rounded, spiracle laterally protruding; articulation with gaster well-defined. Gaster smooth and shiny, with well-marked constriction between abdominal segments III and IV; spiracles on tergites III–V exposed; stridulitrum present on abdominal pretergite IV; abdominal tergum VIII ending in very acute triangle almost forming a spine, cercus about as long as fourth protarsomere, club-shaped, bearing many long hairs issuing from small piligerous tubercles; abdominal sternum IX apically truncate; telomere rounded; penisvalva with ventral margin serrated.
Head covered with scattered, long yellow hairs, longest hairs on posterior part of head; clypeus bearing long, subdecumbent, anteriorly projecting hairs that surpass extended labrum, posterior region of clypeus and frons with shorter appressed yellow hairs. Antenna with many short, appressed and subdecumbent hairs, some longer erect hairs present on first three flagellomere, each about as long as pedicel. Maxillary palp segments III and IV with erect hairs; labrum bearing short erect setae. Dorsum of mesosoma densely covered with yellow decumbent and erect hairs, hairs longer on pronotum and propodeum; pilosity scarce on sides of pronotum and anterior portion of mesopleuron; setae on propodeum long, about same length of mandible. Wings covered with short decumbent hairs. Petiole densely covered with erect, yellow hairs, longer than propodeal hairs. Gaster covered with many subdecumbent yellow hairs.
Head, mesosoma, and petiole brown; scape, pedicel, mandible, labrum, maxilla, labium, legs, and gaster ferruginous brown; flagellum mostly dark brown. Discrete light blue iridescence visible on different body parts depending upon light source used.
See also Tozetto & Lattke (2020).
Karyotype
- n = 53, 2n = 106, karyotype = 18M+88A (Brazil) (Mariano et al., 2004a; Mariano et al., 2015) (Population polymorphism).
- n = 57, 2n = 114 (Brazil) (Mariano et al., 2008) (Population polymorphism).
- n = 58, 2n = 116 (Brazil) (Mariano et al., 2008) (Population polymorphism).
- n = 59, 2n = 118 (Brazil) (Mariano et al., 2008; Mariano et al., 2015) (Population polymorphism).
- n = 60, 2n = 120 (Brazil) (Mariano et al., 2008; Mariano et al., 2015) (Population polymorphism).
- n = 59, 2n = 118 (Brazil) (Santos et al., 2012).
- n = 60 (Brazil) (Santos et al., 2012).
References
- Aguiar, H.J.A.C., Barros, L.A.C., Silveira, L.I., Petitclerc, F., Etienne, S., Orivel, J. 2020. Cytogenetic data for sixteen ant species from North-eastern Amazonia with phylogenetic insights into three subfamilies. Comparative Cytogenetics 14(1): 43–60 (doi:10.3897/CompCytogen.v14i1.46692).
- Barros LAC, Mariano CSF, Pompolo SG and Delabie JHC. 2009. Hsc-FA and NOR bandings on chromosomes of the giant ant Dinoponera lucida Emery, 1901 (Hymenoptera: Formicidae). Comparative Cytogenetics 3:97–102. doi: 10.3897/compcytogen.v3i2.16
- Buys SC, Cassaro R and Salomon D. 2010. Biological observations on Kapala Cameron 1884 (Hymenoptera Eucharitidae) in parasitic association with Dinoponera lucida Emery 1901 (Hymenoptera Formicidae) in Brazil. Tropical Zoology 23:29–34.
- Camargo, K.S. de. 2011. Composicao e diversidade de "Poneromorfas" (Hymenoptera, Formicidae) em duas fitofisionomias de cerrado e padroes de distribuicao de "Poneromorfas", Pseudomyrmecinae e Cephalotini (Myrmicinae) para o Brasil. Thesis, Universidade de Brasilia.
- Campiolo S, Delabie JHC and Agosti D. 2003. Distribuição geográfica: uma ferramenta para avaliação do status de conservação de Dinoponera lucida Emery. Anais do XVI Simpósio de Mirmecologia, UFSC, Florianópolis –SC, pp. 359–360.
- Cardoso, D. C., Cristiano, M. P. 2021. Karyotype diversity, mode, and tempo of the chromosomal evolution of Attina (Formicidae: Myrmicinae: Attini): Is there an upper limit to chromosome number? Insects 1212, 1084 (doi:10.3390/insects12121084).
- Correia, J.P.S.O., Mariano, C S F., Delabie, J.H.C., Lacau, S., Costa, M.A. 2016. Cytogenetic analysis of Pseudoponera stigma and Pseudoponera gilberti (Hymenoptera: Formicidae: Ponerinae): a taxonomic approach. Florida Entomologist 99: 718-721.
- Curbani, F., Zocca, C., Ferreira, R.B., Waichert, C., Sobrinho, T.G., Srbek-Araujo, A.C. 2021. Litter Surface Temperature: A Driving Factor Affecting Foraging Activity in Dinoponera lucida (Hymenoptera: Formicidae). Sociobiology 68: e-6030 (doi:10.13102/sociobiology.v68i1.6030).
- Dias, A.M., Lattke, J.E. 2021. Large ants are not easy – the taxonomy of Dinoponera Roger (Hymenoptera: Formicidae: Ponerinae). European Journal of Taxonomy 784, 1–66 (doi:10.5852/ejt.2021.784.1603).
- Disney, R. H. L., M. A. L. Braganca, and M. C. Teixeira. 2015. New Species of Scuttle Flies (Diptera: Phoridae) Associated with a Ponerine Ant (Hymenoptera: Formicidae) in Brazil. Sociobiology. 62:124-127. doi:10.13102/sociobiology.v62i1.124-127
- Emery, C. 1901b. Notes sur les sous-familles des Dorylines et Ponérines (Famille des Formicides). Ann. Soc. Entomol. Belg. 45: 32-54 (page 48, worker described)
- Escarraga, M. E., J. E. Lattke, and C. O. Azevedo. 2017. Discovery of the Dinoponera lucida male (Hymenoptera, Formicidae), a threatened giant ant from the Atlantic rain forest. Zootaxa. 4347:128-136. doi:10.11646/zootaxa.4347.1.7
- Esteves, F.A., Fisher, B.L. 2021. Corrieopone nouragues gen. nov., sp. nov., a new Ponerinae from French Guiana (Hymenoptera, Formicidae). ZooKeys 1074, 83–173 (doi:10.3897/zookeys.1074.75551).
- Kempf, W. W. 1971. A preliminary review of the ponerine ant genus Dinoponera Roger (Hymenoptera: Formicidae). Stud. Entomol. 14: 369-394 (page 376, Raised to species)
- Lenhart, P.A., Dash, S.T. & Mackay, W.P. 2013. A revision of the giant Amazonian ants of the genus Dinoponera (Hymenoptera, Formicidae). Journal of Hymenoptera Research 31, 119–164.
- Lorite, P., Palomeque, T. 2010. Karyotype evolution in ants (Hymenoptera: Formicidae), with a review of the known ant chromosome numbers. Myrmecological News 13: 89-102.
- Mariano CSF, Pompolo SDG, Campos Barros LA, Mariano-Neto E, Campiolo S and Delabie JHC. 2008. A biogeographical study of the threatened ant Dinoponera lucida Emery (Hymenoptera: Formicidae: Ponerinae) using a cytogenetic approach. Insect Conservation and Diversity 1: 161–168. doi: 10.1111/j.1752-4598.2008.00022.x
- Mariano, C. S. F.; Delabie, J. H. C.; Ramos, L. S.; Lacau, S.; Pompolo, S. G. 2004. Dinoponera lucida Emery (Formicidae: Ponerinae): the highest number of chromosomes known in Hymenoptera. Naturwissenschaften 91: 182-185 (page 182-185, karyotype described)
- Mariano, C.S.F., Santos, I.S., Silva, J.G., Costa, M.A., Pompolo, S.G. 2015. Citogenética e evolução do cariótipo em formigas poneromorfas. In: Delabie, J.H.C., Feitosa, R.M., Serrao, J.E., Mariano, C.S.F., Majer, J.D. (eds) As formigas poneromorfas do Brasil, 1st edn. Ilhéus, Brasil, pp 102–125 (doi:10.7476/9788574554419.0010).
- Marques-Silva S, Matiello-Guss CP, Delabie JHC, Mariano CSF, Zanuncio JC and Serrão JE. 2006.Sensilla and secretory glands in the antennae of a primitive ant: Dinoponera lucida (Formicidae:Ponerinae). Microscopy Research and Technique 69: 885–890. doi: 10.1002/jemt.20356
- Peixoto AV, Campiolo S, Lemes TN, Delabie JHC and Hora RR. 2008. Comportamento e estrutura reprodutiva da formiga Dinoponera lucida Emery (Hymenoptera, Formicidae). Revista Brasileira de Entomologia 52: 88–94. doi: 10.1590/S0085-56262008000100016
- Ramirez-Esquivel, F., Zeil, J., Narendra, A. 2014. The antennal sensory array of the nocturnal bull ant Myrmecia pyriformis. Arthropod Structure, Development 43, 543–558. (doi:10.1016/j.asd.2014.07.004).
- Richter, A., Boudinot, B.E., Hita Garcia, F., Billen, J., Economo, E.P., Beutel, R.G. 2023. Wonderfully weird: the head anatomy of the armadillo ant, Tatuidris tatusia (Hymenoptera: Formicidae: Agroecomyrmecinae), with evolutionary implications. Myrmecological News 33: 35-75 (doi:10.25849/MYRMECOL.NEWS_033:035).
- Santos, I.S., Delabie, J.H.C., Silva, J.G., Costa, M.A., Barros, L.A.C., Pompolo, S.G. & Mariano, C.S.F. 2012. Karyotype differentiation among four Dinoponera (Formicidae: Ponerinae) species. Florida Entomologist 95(3), 737-742
- Serrão JE, Castro RCA, Zanuncio JC, Mariano CSF and Delabie JHC. 2009. Epidermal glands in the abdomen of a basal ant Dinoponera lucida (Formicidae: Ponerinae). Microscopy Research and Technique 72: 28–31. doi: 10.1002/jemt.20641
- Silva, P.S., Koch, E.B. de A., Arnhold, A., Delabie, J.H.C. 2022. Review of distribution modeling in ant (Hymenoptera: Formicidae) biogeographic studies. Sociobiology 69(4), e7775 (doi:10.13102/sociobiology.v69i4.7775).
- Simon, S., Schoereder, J.H., Teixeira, M.D.C. 2020. Environmental response of Dinoponera lucida Emery 1901 (Hymenoptera: Formicidae), an endemic threatened species of the Atlantic Forest Central Corridor. Sociobiology 67(1), 65-73 (doi:10.13102/sociobiology.v67i1.3662).
- Tozetto, L., Lattke, J.E. 2020. Revealing male genital morphology in the giant ant genus Dinoponera with geometric morphometrics. Arthropod Structure & Development 57, 100943 (doi:10.1016/j.asd.2020.100943)).
- Troya, A., Marcineiro, F., Lattke, J.E. & Longino, J. 2022. Igaponera curiosa, a new ponerine genus (Hymenoptera: Formicidae) from the Amazon. European Journal of Taxonomy 823: 82–101 (doi:10.5852/ejt.2022.823.1817).
- Zocca, C., Curbani, F., Ferreira, R.B., Weichert, C., Sobrinho, T.G., Srbek-Araujo, A.C. 2021. A day in the life of the giant ant Dinoponera lucida Emery, 1901 (Hymenoptera, Formicidae): Records of activities and intraspecific interactions. Sociobiology 68, 6166 (doi:10.13102/sociobiology.v68i2.6166).
References based on Global Ant Biodiversity Informatics
- Borgmeier T. 1923. Catalogo systematico e synonymico das formigas do Brasil. 1 parte. Subfam. Dorylinae, Cerapachyinae, Ponerinae, Dolichoderinae. Archivos do Museu Nacional (Rio de Janeiro) 24: 33-103.
- Dos Santos Ferreira Mariano C., S. Das Gracas Pompolo, L. A. Campos Barros, E. Mariano-Neto, S. Campiolo, and J. H. C. Delabie. 2008. A biogeographical study of the threatened ant Dinoponera lucida Emery (Hymenoptera: Formicidae: Ponerinae) using a cytogenetic approach. Insect Conservation and Diversity 1: 161168.
- Emery C. 1911. Hymenoptera. Fam. Formicidae. Subfam. Ponerinae. Genera Insectorum 118: 1-125.
- Kempf W. W. 1971. A preliminary review of the ponerine ant genus Dinoponera Roger (Hymenoptera: Formicidae). Studia Entomologica 14: 369-394.
- Kempf W. W. 1978. A preliminary zoogeographical analysis of a regional ant fauna in Latin America. 114. Studia Entomologica 20: 43-62.
- Kempf, W.W. 1972. Catalago abreviado das formigas da regiao Neotropical (Hym. Formicidae) Studia Entomologica 15(1-4).
- Lenhart, P. A.; Dash, S. T.; and Mackay, W. P. 2013. A revision of the giant Amazonian ants of the genus Dinoponera (Hymenoptera, Formicidae). Journal of Hymenoptera Research 31: 119-164
- Peixoto, A.V., S. Campiola, T.N. Lemes, J.H.C. Delabie and R.R. Hora. 2008.Comportamento e estrutura reprodutiva da formiga Dinoponera lucida Emery (Hymenoptera, Formicidae). Revista Brasileira de Entomologia 52(1): 88-94
- Vieira Peixoto A., S. Campiolo, and J. H. C. Delabie. 2010. Basic ecological information about the threatened ant, Dinoponera lucida Emery (Hymenoptera: Formicidae: Ponerinae), aiming its effective long-term conservation. In: Species Diversity and Extinction, Chapter 5, Tepper G. H. eds. pp. 183-213.
- Pages using DynamicPageList3 parser function
- IUCN Red List vulnerable species
- Tropical
- Eucharitid wasp Associate
- Host of Kapala sp.
- Phorid fly Associate
- Host of Apocephalus exlucida
- Host of Megaselia sp. A
- Host of Megaselia sp. B
- Karyotype
- Species
- Extant species
- Formicidae
- Ponerinae
- Ponerini
- Dinoponera
- Dinoponera lucida
- Ponerinae species
- Ponerini species
- Dinoponera species
- Ssr



