Leptanilla japonica
| Leptanilla japonica | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Leptanillinae |
| Tribe: | Leptanillini |
| Genus: | Leptanilla |
| Species: | L. japonica |
| Binomial name | |
| Leptanilla japonica Baroni Urbani, 1977 | |
| Common Name | |
|---|---|
| Yamato-mukashi-ari | |
| Language: | Japanese |
Leptanilla japonica nests in small hollows in the soil (Japanese Ant Image Database). Wong and Guérnard (2016) report a single worker of a Leptanilla species identified as Leptanilla sp. cf. japonica from Hong Kong. The specimen was collected in leaf litter using the Winkler extractor method in Lung Fu Shan Park (22°16.823'N, 114°8.270'E, 116m), located on Hong Kong Island on November 25th 2014. CASENT0914941. Photographs of the specimen are available on Antweb.org. While the specimen appears to match the description and characteristics of Leptanilla japonica, we are hesitant to formerly identify it as such due to the important disjunction observed in the distribution of this species. To the best of our knowledge this species has been reported only from Honshu Island in Japan (Antmaps 2015) and it is thus surprising to find it so far south (≥2500km) in the absence of other intermediate records. Future surveys in Asia should reveal if this record represents a valid record of L. japonica or a new cryptic species morphologically similar to the latter.
| At a Glance | • Larval Hemolymph Feeding |
Identification
Ogata et. al. (1995) - The female of this species is unique in having eyes. The male is also characteristic in having hairy eyes, small projections on the parameres and a simple hypopygium which is not bifurcate apically.
Japanese Ant Image Database: Similar to Leptanilla morimotoi but distinguished in having 4 teeth on the mandible (where morimotoi have 3). The second tooth, counting from the apex, is sometimes small, low and obscure.
Keys including this Species
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: 35.15000153° to 35.13333333°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Palaearctic Region: Japan (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
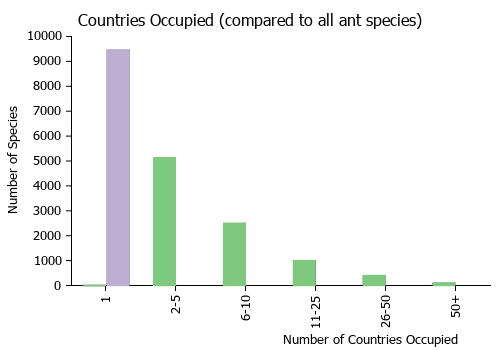
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
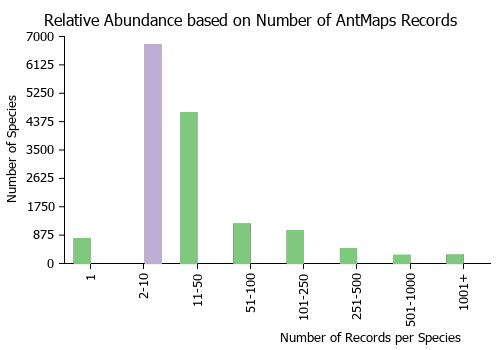
|
Biology
The following observations are provided by Masuko (1989):
The larvae of the migratory Japanese ant Leptanilla japonica Baroni Urbani have a specialized duct organ on each side of the 4th abdominal segment. Behavioral and histological studies have shown that the adult ants are able to imbibe hemolymph directly from the larval body cavity through these organs, each of which is referred to here as a "larval hemolymph tap." Laboratory observations further confirm that larval hemolymph feeding (LHF) is the sole source of nutrient for the queens. L. japonica is cyclical in its brood production. All larvae in a colony develop in concert, and when they are mature, the queen performs much active LHF on them. Nourished in this way, she achieves full physogastry within a period of only a few days and then lays a batch of 100-200 eggs. LHF thus facilitates synchronization of brood maturation and concentrated production of eggs by the queen. The larval hemolymph tap has probably evolved de novo in Leptanilla, in relation to cyclical brood production by species with small colony populations. Obviously, the function of this larval organ is non-selfish, contributing to overall colony-level functioning. The larvae of Leptanilla are thus properly qualified as a distinct class of caste in ant societies.
Larval hemolymph feeding in Leptanilla
This behavior was first noted during observation of the queen of a colony. After briefly but strongly stroking a final-instar larva with her antennae, she held it with her legs and attached her lower mouthparts to its lateral surface. She then maintained oral contact with the lateral body surface while moving the larva with her forelegs, as if searching for some specific spot on its body. During this exploration, the queen frequently nipped the larva lightly with her mandibles, concentrating especially on the thin lateral borders of the abdominal segments. The queen grasped the larva with all three leg pairs, usually lying on her side during this act. Finally, she ceased exploration at the 4th abdominal segment and attached her mouthparts to it for a period of time. During culture of this colony, the queen was never observed to feed on prey, even though the larvae and workers were actively feeding on prey at this time. The frequency of this behavior of the L. japonica queen increased greatly when she became physogastric. Shortly thereafter she laid a batch of about 50 eggs within a period of only 3 days (3 6 April 1982). This colony had a biased composition due to damage caused by field collection; the colony consisted of only 12 larvae and approximately 120 workers. It perished 43 days after the onset of laboratory rearing. Further behavioral details were studied using colony 83-14, which had a more normal composition (1 queen, approx. 130 workers, and 100 larvae). During the 103-day life of this colony, feeding on prey by the queen was never observed, although the ants were provided with 25 prey items (intact geophilomorph centipedes or their chopped bodies), most of which were consumed avidly by larvae and workers. These observations were similar to the larval hemolymph feeding previously discovered by the author in Amblyopone silvestrii (Masuko 1986).
To determine whether the 4th abdominal segment is a region involved in LHF behavior and to analyze the behavioral characteristics of Leptanilla japonica queens, a behavioral time budget was developed for this queen. Her activities were assayed for a total of 745 rain, comprising eight 90-100 min observation periods, conducted from 12 January to 13 March 1983. This period covered both her nonphysogastric and fully physogastric conditions. During these observations, the queen performed a total of 67 acts of "freezing", i.e. the mouthparts were attached to different larval regions for varying lengths of time. On 59 occasions the region involved was recorded; on 51 occasions time spent at that region was accurately measured (Fig. 2). The results show clearly that the 4th abdominal segment is a region of special emphasis for attention activity, in terms of both its frequency and duration. The frequency of attention to the abdominal tip, where the larval hindgut opens, was also high, but the mean durations observed indicate no persistence by the queen in this region (see also Larval duct organs).
Evolution of LHF in Leptanilla.
LHF in Stigmatomma silvestrii has been suggested to be a nutritive adaptation related to the absence of other modes of social food transfer and to the specialized predation on large sporadic prey (centipedes) in this species (Masuko 1986). Only mature or near-mature larvae of the final (5th) instar are employed for LHF by S. silvestrii queens. Since these larvae are present in colonies throughout the active season of the year, through LHF the queen's fecundity can be sustained during unstable food conditions, thus avoiding destruction of the larvae by the only possible alternative, cannibalism. Laboratory and field data strongly suggest that Leptanilla is also a specialized predator of geophilomorph centipedes (Masuko et al., unpublished data). Leptanilla also lacks the habit of oral trophallaxis, and feeding the queens with trophic eggs produced by workers is not possible because due to the absence of ovaries workers cannot produce eggs. Prey is returned to the nest in Stigmatomma by solitary foragers; in contrast, Leptanilla workers are, apparently, obligate group-predators using trail systems, though on a far smaller scale than is known among the army ants (members of the Dorylinae and Ecitoninae). Field data on hunting success are lacking for both Leptanilla and Stigmatomma. Nevertheless, considering the much smaller body size of Leptanilla workers (1.2 mm vs 4-5 mm in Stigmatomma), and that group foraging is supported by a very small worker force (the total observed worker population in L. japonica colonies is only 100-200), it is possible that prey acquisition in Leptanilla is more limited than in Stigmatomma. Furthermore, the nest emigration observed in Leptanilla could be related to local food depletion, which has been invoked to account for the nomadism of army ants (Wilson 1971; Topoff 1984). The selective advantage of LHF previously suggested for Stigmatomma could, therefore, hold also for Leptanilla. Efficient and opportune intranidal distribution of food is especially important in ant species that produce a large amount of brood cyclically and in which brood development is synchronized. Typical of this are Leptanilla, the army ants and some ponerine species belonging to Cerapachys (Holldobler 1982), Simopelta (Gotwald and Brown 1966), and Onychomyrmex (Taylor, cited in Schneirla 1971, p. 167). As previously suggested (Masuko 1987), LHF is an expedient way for the queen to synchronize mass egg-production with the maturation of larvae in her colony. Nothing is known in regard to the nutrition of the queens in army ants (Rettenmeyer 1963; Schneirla 1971; Gotwald 1982). Cannibalism of mature brood, either destructively or in a nondestructive manner like LHF, are likely modes of queen nutrition in these ants. Worker trophic eggs are another potential source of nourishment for the queens, but their production has not been confirmed in army ants.
According to the descriptions of ant larvae by Wheeler and Wheeler (e.g., 1976 and 1984), structures like the duct organs of Leptanilla are not known for dorylines or ecitonines. Hence, I argue the development of duct organs in Leptanilla as a feature related to small colony size.
Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- japonica. Leptanilla japonica Baroni Urbani, 1977c: 460, figs. 13, 312 (w.) JAPAN. Wheeler, G.C. & Wheeler, J. 1989c: 186 (l.); Ogata, Terayama & Masuko, 1995: 27 (q.m.). See also: Masuko, 1987: 597; Masuko, 1990: 31.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description

Queen
Ogata et. al. (1995) - HW (head width) 0.25 mm, HL (head length) 0.33 mm, Cl (cephalic index: HW x 100/HL) 75.8, SL (scape length) 0.13mm, PW (pronotal width) 0.21 mm, WL (Weber's length of trunk) 0.54mm, PNL (petiolar node length) 0.13mm, PNW (petiolar node width) 0.16mm, TL (total length) 1.8 mm (1 individual measured).
Head subrectangular with concave posterior margin and subparallel sides; posterior corners rounded. Mandibles stout, edentate. Clypeus with transverse anterior margin. Antennal insertion situated close to anterior margin of head. Antennae 12-segmented; scape short and robust; pedicel longer than broad, separated from the rest of funiculus by constriction; antennal segments 3-11 each broader than long; apical segment longer than two preceding segments together. Eyes present, each consisting of two facets, situated on the midlength of sides of head. Ocelli absent.
Trunk flattened; sides subparallel in dorsal view, nearly 3x as long as broad. Pronotum without anterior corners in dorsal view; posterior margin straight. Propleuron large. Metanotal groove absent. Metapleural gland distinct. Propodeum with distinct spiracle situated above hind coxal base. Middle leg with single small tibial spur; hind leg with one larger pectinate spur and small simple spur. Petiolar node broader than long, about l.5x as broad as long, narrower than trunk; oval in dorsal view. Ventral portion of petiole produced, with distinct anterior process. Gaster large; first gastral segment about 1.3x as broad as long. Body colour light reddish yellow. Hairs short, abundant on gaster.
Male
Ogata et. al. (1995) - HW 0.19, HL 0.25, CI 75, PW 0.19, WL 0.43, forewing L 1.06, hindwing L 0.60, PNL 0.11, PNW 0.08, TL 1.30 (1 individual measured).
Head longer than broad, with rounded posterior corners and subparallel sides; posterior margin slightly emarginate without occipical carina. Mandibles reduced, non-opposable, forming small rounded and flattened lobes with concave dorsal surface. Palp formula 1,1. Clypeus narrow, shallowly depressed and fused to cranium posteriorly; anterior margin almost straight. Antennae 13-segmented; scape short and stout, reaching posteriormost portion of eye; funiculus filiform, each segment subglobose and slightly longer than broad except for apical one; apical segment longest, nearly as long as two preceding segments together. Eyes hairy, large and convex, situated anteriorly. Ocelli developed.
Trunk elongate. Pronotum long, nearly twice as long as propleuron; anterior projection thin, not forming cercival collar; posterolateral portion concave; both sides close to each other at posteroventral portion. Propleuron short, very convex ventrally. Mesoscutum elongate, about half as long as trunk; dorsal surface arched, without notauli and parapsidal furrows. Mesoscutellum slightly raised, not overhanging metanotum. Metanotum small, forming short transverse sclerite. Oblique furrow on mesepisternum weakly developed. Propodeum cylindrical and marginate posteriorly, with arched node. Metapleural gland absent. Middle leg with a single small spur; hind leg with two small spurs.
Forewing with weakly developed vein Sc + R + Rs; other veins and pterostigma absent. Hindwing narrow without any veins.
Petiole subglobous, constricted posteriorly, without anterior peduncle. Cerci absent.
Hypopygium small, short and broad, with rounded apical projection which is low and never bifurcate. Genitalia not retractile; basal ring small and thin, weakly sclerotized and fused to hypopygium ventrally; basal portion of parameres widely separated dorsally, but contiguous to each other ventrally; apical portion of parameres extended dorsally, forming flattened apices each with one outer and two inner teeth; digitus small cuspis absent; aedeagal plate flattened, dorsally projecting without serrate ventral margin.
Type Material
- Paratype, 2 workers, Manazuru, Kanagawa Prefect, Japan, Kubota,M., ANIC32-017902, Australian National Insect Collection.
References
- Ogata K., Terayama, M. & Masuko, M. 1995. The ant genus Leptanilla: discovery of the worker-associated male of L. japonica, and a description of a new species from Taiwan (Hymenoptera: Formicidae: Leptanillinae). Systematic Entomology 20: 27-34. (page 27, queen, male described)
- Baroni Urbani, C. 1977c. Materiali per una revisione della sottofamiglia Leptanillinae Emery (Hymenoptera: Formicidae). Entomol. Basil. 2: 427-488 (page 460, figs. 13, 312 worker described)
- Bolton, B. 1995b. A new general catalogue of the ants of the world. Cambridge, Mass.: Harvard University Press, 504 pp. (page 229, catalogue)
- Cantone S. 2017. Winged Ants, The Male, Dichotomous key to genera of winged male ants in the World, Behavioral ecology of mating flight (self-published).
- Griebenow, Z. 2020. Delimitation of tribes in the subfamily Leptanillinae (Hymenoptera: Formicidae), with a description of the male of Protanilla lini Terayama, 2009. Myrmecological News 30: 229-250. (doi:10.25849/MYRMECOL.NEWS_030:229).
- Griebenow, Z. 2024. Systematic revision of the ant subfamily Leptanillinae (Hymenoptera, Formicidae). ZooKeys 1189, 83–184 (doi:10.3897/zookeys.1189.107506).
- Griebenow, Z.H., Isaia, M., Moradmand, M. 2022. A remarkable troglomorphic ant, Yavnella laventa sp. nov. (Hymenoptera: Formicidae: Leptanillinae), identified as the first known worker of Yavnella Kugler by phylogenomic inference. Invertebrate Systematics 36(12), 1118-1138 (doi:10.1071/is22035).
- Hashimoto, Y. 1990. Unique features of sensilla on the antennae of Formicidae (Hymenoptera). Applied Entomology and Zoology 25: 491-501.
- Ito, F., Hashim, R., Mizuno, R., Billen, J. 2021. Notes on the biology of Protanilla sp. (Hymenoptera, Formicidae) collected in Ulu Gombak, Peninsular Malaysia. Insectes Sociaux (doi:10.1007/s00040-021-00839-z).
- Ito, F., Yamane, S. 2020. Behavior of the queen of Leptanilla clypeata Yamane et Ito collected in the Bogor Botanical Gardens, West Java, Indonesia (Hymenoptera; Formicidae), with a note on colony composition and a description of the ergatoid queen. Asian Myrmecology 12, e12004 (doi:10.20362/am.012004).
- Makuko, K. 1989. Larval hemolymph feeding in the ant Leptanilla japonica by use of a specialized duct organ, the "larval hemolymph tap" (Hymenoptera: Formicidae). Behavioral Ecolology and Sociobiology 24:122-132.
- Masuko, K. 1987. Leptanilla japonica: the first bionomic information on the enigmatic ant subfamily Leptanillinae. Pp. 597-598 in: Eder, J., Rembold, H. (eds.) Chemistry and biology of social insects. München: Verlag J. Peperny, xxxv + 757 pp. (page 597, see also)
- Masuko, K. 1990a. Behavior and ecology of the enigmatic ant Leptanilla japonica Baroni Urbani (Hymenoptera: Formicidae: Leptanillinae). Insectes Soc. 37: 31-57 (page 31, see also)
- Meurville, M.-P., LeBoeuf, A.C. 2021. Trophallaxis: the functions and evolution of social fluid exchange in ant colonies (Hymenoptera: Formicidae). Myrmecological News 31: 1-30 (doi:10.25849/MYRMECOL.NEWS_031:001).
- Qian, Y.-H., Zheng-Hui, X., Man, P., Liu, G.-L. 2024. Three new species of the ant genus Leptanilla (Hymenoptera: Formicidae) from China, with a key to the world species. Myrmecological News 34: 21-44 (doi:10.25849/MYRMECOL.NEWS_034:021).
- Saroj, S., Mandi, A., Dubey, A.K. 2022. A new species of the rare ant genus, Leptanilla Emery (Hymenoptera: Formicidae) from Eastern Himalaya, India. Asian Myrmecology 15, e015005 (doi:10.20362/am.015005).
- Wheeler, G. C.; Wheeler, J. 1989c [1988]. The larva of Leptanilla japonica, with notes on the genus (Hymenoptera: Formicidae: Leptanillinae). Psyche (Camb.) 95: 185-189 (page 186, larva described)
- Wong, M.K.L., Guénard, B. 2016. Leptanilla hypodracos sp. n., a new species of the cryptic ant genus Leptanilla (Hymenoptera, Formicidae) from Singapore, with new distribution data and an updated key to Oriental Leptanilla species. ZooKeys. 551:129–144. (doi: 10.3897/zookeys.551.6686).
- Yamada, A., Van Dang, A., Eguchi, K. 2023. Natural history notes of the rare enigmatic ant Opamyrma hungvuong: A first glimpse of their preying behavior on centipedes (Hymenoptera: Formicidae: Leptanillinae). Asian Myrmecology 16, e016009 (doi:10.20362/am.016009).
References based on Global Ant Biodiversity Informatics
- Huong N. T. T., P. V. Sang, and B. T. Viet. 2015. A preliminary study on diversity of ants (Hymenoptera: Formicidae) at Hon Ba Nature Reserve. Environmental Scientific Conference 7: 614-620.
- Ogata K.; Terayama, M.; Masuko, K. 1995. The ant genus Leptanilla: discovery of the worker-associated male of L. japonica, and a description of a new species from Taiwan (Hymenoptera: Formicidae: Leptanillinae). Systematic Entomology 20:27-34.



