Leptogenys gaigei
| Leptogenys gaigei | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Ponerinae |
| Tribe: | Ponerini |
| Genus: | Leptogenys |
| Species group: | luederwaldti |
| Species: | L. gaigei |
| Binomial name | |
| Leptogenys gaigei Wheeler, W.M., 1923 | |
| Synonyms | |
| |
Reproduction by gamergates is suspected for this species given the abundant material available for study, including nest series, but not one recognizable queen as of yet. A similar mode of reproduction might be possible for Leptogenys cuneata, the putative sister species of Leptogenys gaigei. (Lattke 2011)
Identification
Lattke (2011) - A member of the luederwaldti species group. Head elongate with flattened eyes at cephalic mid-length; apical strigil of protibia with lateral translucent lobe; propodeum with blunt triangular lobes; petiolar node in lateral view triangular with convex anterodorsal margin; body smooth and shining, mostly black with blue opalescence.
Keys including this Species
Distribution
It is known from Amazon-Orinoco watershed Ecuador, Brazil, Suriname, Colombia, and French Guiana.
Latitudinal Distribution Pattern
Latitudinal Range: 1.839722222° to -7.730534°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: Brazil, Colombia, Ecuador, French Guiana, Guyana (type locality), Suriname.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
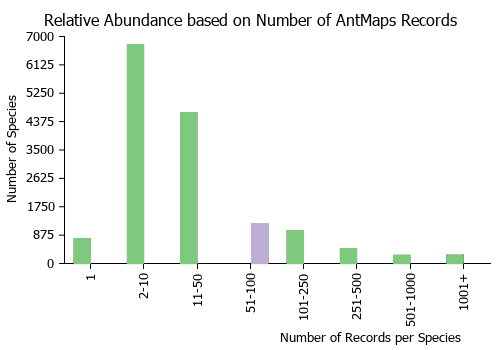
|
Biology
|
Castes
Queen unknown.
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- gaigei. Leptogenys (Lobopelta) gaigei Wheeler, W.M. 1923d: 7, fig. 2 (w.) GUYANA. Lattke, 2011: 184 (m.). Senior synonym of defuga, melzeri: Lattke, 2011: 183.
- defuga. Leptogenys (Lobopelta) gaigei var. defuga Wheeler, W.M. 1923d: 8 (w.) GUYANA. Junior synonym of gaigei: Lattke, 2011: 183.
- melzeri. Leptogenys (Lobopelta) melzeri Borgmeier, 1930: 27, pl. 4, figs. 15, 20, 22 (w.) BRAZIL. Junior synonym of gaigei: Lattke, 2011: 183.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Wheeler (1923) describes L. gaigei var. defuga, on account of a shinier cephalic dorsum due to the more distanced and shallower punctures, and lessened pubescence and pilosity as well. Such differences can be seen in other Leptogenys species, and both L. gaigei and L. defuga share a transparent lobe next to the protibial strigil. No characters could be found to justify keeping the two forms as distinct entities. Borgmeier (1930) suspected L. melzeri was close to L. gaigei, though he compared L. melzeri with Leptogenys luederwaldti. L. melzeri shares with L. gaigei the transparent lobe next to the protibial strigil. The eye in L. melzeri occupies at least one-third of the lateral cephalic margin when seen in full-face view, and the mesonotum is about as long as broad in dorsal view. It is also larger in dimensions than most L. gaigei, but the Pará series are as large as the L. melzeri forms. The promesonotal margin broadly is convex, and is higher than the propodeal margin in lateral view. The size of the propodeal lobes is variable. The type of L. melzeri was collected in Campinas, Goias by P. Schwarzmaier on 6.ix.1928, and deposited in the Instituto Biologico, Rio de Janeiro as specimen 4710 (Borgmeier 1930). Besides the holotype, abundant material from the type locality, collected by Schwarzmaier, and from roughly the same time period when the type was collected, was available for study in the Museu de Zoologia da Universidade de Sao Paulo, all of which corresponded very close to the detailed description of L. melzeri. Leptogenys cuneata shares with L. gaigei the translucent lobe next to the protibial strigil but can be distinguished by its larger size, total absence of propodeal lobes, and the very elongate petiolar node with a mostly straight anterodorsal margin in lateral view.
Description
Worker
Metrics (n = 7): HL 1.16 – 1.44; HW 0.76 – 0.91; ML 0.46 – 0.58; EL 0.23 – 0.30; SL 1.14 – 1.52; PW 0.08 – 0.78; WL 1.77 – 2.23; PH 0.63 – 0.73; PL 0.66 – 0.89; DPW 0.38 – 0.46 mm. CI 0.63 – 0.65; MI 0.55 – 0.66; OI 0.29 – 0.33; SI 1.50 – 1.67; LPI 0.80 – 0.96; DPI 0.48 – 0.58.
Head rectangular in full-face view, slightly wider anterad than posterad; posterior margin straight; lateral margin broadly convex; anterior clypeal margin evenly tapering to triangular median lobe, apex pointed or rounded. Eye mostly flattened, weakly convex, set slightly dorsad, approximately at mid-length of lateral cephalic margin, eye occupies between one-fourth and one-third of lateral cephalic margin. Scape surpasses posterior cephalic margin by distance more than one-third its length, scape thickest at basal third, thinner apically; mostly smooth and shining with piligerous punctulae. Second antennal segment less than one-third length of third segment, third segment subequal in length to fourth and fifth combined; funicular segments subcylindrical. Cephalic dorsum mostly smooth and shining with sparse to dense piligerous punctulae; clypeus with low rugosities. Cephalic ventral surface mostly smooth and shining, discal area devoid of pubescence. External maxilla and labium mostly smooth and shining with weak strigulae. PF 4,4. Mandible shuts tight against clypeus, mandible of uniform width, basal margin sinuate, masticatory border edentate, separated from basal margin by single denticle, dorsum mostly smooth and shining except for weak strigulae, basal groove strongly impressed; convexity of basal margin with row of 3 prominent hairs.
Mesosoma in lateral view with fairly straight dorsal margin, promesonotum forming broad convexity slightly higher than dorsal propodeal margin, metanotal groove distinct but shallow, propodeal declivity with low lobe at spiracular height, lobe usually jagged on dorsal margin. Pronotum smooth and shining with sparse piligerous punctae; ventral sulcus weakly impressed; mesosomal sides mostly smooth and shining, some rugulae present along ventral margin of mesometapleura. Mesopleuron rectangular; mesometapleural suture scrobiculate; metapleural-propodeal suture absent to weakly impressed as series of irregular depressions more or less in a row, propodeal spiracle oval and facing posterolaterally; metathoracic spiracle crescent shaped, spiracular prominence flattened to slightly convex, round to oval. Mesonotum transverse in dorsal view, rectangular, posterior margin anteriorly convex; propodeal declivity with transverse strigae, median area concave; propodeum with low triangular blunt lobe to none at all. Mesosternal process with posterior margin angular to rounded in lateral view, minute posterior spine sometimes present.
Petiole in lateral view triangular with convex anterodorsal margin and vertical to slightly convex posterior margin, smooth and shining; elongate in dorsal view with anterior margin less than half as wide as posterior margin, lateral margin concave. Postpetiole with convex anterior margin; gaster smooth and shining with sparse piligerous punctulae; pygidial dorsal margin broadly convex in lateral view. Procoxa mostly smooth and shining in lateral view, with sparse punctulae; protibial apex with slender and fine seta just anterad of strigil, at least one-third length of strigil, seta missing in some specimens; strigil with basal translucent triangular lobe; setae at apical margin of tarsi reduced. Scape with abundant subdecumbent pilosity. Scape, mandible, legs, and gastral apex dark brown; funiculus ferruginous brown; rest of body black with blue opalescence.
Male
Cephalic dorsum mostly smooth and shining with sparse piligerous punctulae; pronotum laterally with low rugosities to striae, prosternum with low rugosities; notaulus well impressed, scrobiculate, joining posterad and extending to posterior scutal margin. Shallow straight sulcus extends longitudinally from posterolateral scutum to mid distance of lateral scutum. Protibial apex lacking transparent lobe. Most of head, thorax, and abdomen brown; clypeus, and most of antennal segments III – XIII light brown; legs, mouthparts, antennal segments I – II, and narrow fringe at each end of antennal segments III – XIII yellow to brown yellow.
Type Material
Lattke (2011):
Leptogenys (Lobopelta) gaigei. Syntype workers: Guiana (= British Guiana), Dunoon, 17. – 20.viii.1914 (F.M. Gaige) (Museum of Comparative Zoology) [examined].
Leptogenys (Lobopelta) gaigei var. defuga. Syntype workers: Guiana (= British Guiana), Kartabo, 12.viii. 1920, nest under old log (W.M. Wheeler) (MCZC) [examined].
Leptogenys (Lobopelta) melzeri. Holotype worker: Brazil, Goias, Campinas, 6.x.1928, Cat. No. 4701 (P.J.S. Schwarzmaier) (IBSP) [examined].
References
- Albuquerque, E., Prado, L., Andrade-Silva, J., Siqueira, E., Sampaio, K., Alves, D., Brandão, C., Andrade, P., Feitosa, R., Koch, E., Delabie, J., Fernandes, I., Baccaro, F., Souza, J., Almeida, R., Silva, R. 2021. Ants of the State of Pará, Brazil: a historical and comprehensive dataset of a key biodiversity hotspot in the Amazon Basin. Zootaxa 5001, 1–83 (doi:10.11646/zootaxa.5001.1.1).
- Camargo, K.S. de. 2011. Composicao e diversidade de "Poneromorfas" (Hymenoptera, Formicidae) em duas fitofisionomias de cerrado e padroes de distribuicao de "Poneromorfas", Pseudomyrmecinae e Cephalotini (Myrmicinae) para o Brasil. Thesis, Universidade de Brasilia.
- Franco, W., Ladino, N., Delabie, J.H.C., Dejean, A., Orivel, J., Fichaux, M., Groc, S., Leponce, M., Feitosa, R.M. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674, 509–543 (doi:10.11646/zootaxa.4674.5.2).
- Lattke, J.E. 2011. Revision of the New World species of the genus Leptogenys Roger (Insecta: Hymenoptera: Formicidae: Ponerinae). Arthropod Systematics & Phylogeny. 69:127-264.
- Wheeler, W. M. 1923d. Report on the ants collected by the Barbados-Antigua Expedition from the University of Iowa in 1918. Stud. Nat. Hist. Iowa Univ. 10(3 3: 3-9 (page 7, fig. 2 worker described)
References based on Global Ant Biodiversity Informatics
- Alonso L. E., J. Persaud, and A. Williams. 2016. Biodiversity assessment survey of the south Rupununi Savannah, Guyana. BAT Survey Report No.1, 306 pages.
- Borgmeier T. 1930. Duas rainhas de Eciton e algumas outras formigas brasileiras. Archivos do Instituto Biológico (São Paulo) 3: 21-40.
- Carvalho Pereira L. P. 2012. Estrutura da comunidade de formigas poneromorfas (Hymenoptera:Formicidae) em uma área da Floresta Amazônica. Master Thesis Universidade Federal Rural do Rio de Janeiro. 64 pages.
- Fernández F., and T. M. Arias-Penna. 2008. Las hormigas cazadoras en la región Neotropical. Pp. 3-39 in: Jiménez, E.; Fernández, F.; Arias, T.M.; Lozano-Zambrano, F. H. (eds.) 2008. Sistemática, biogeografía y conservación de las hormigas cazadoras de Colombia. Bogotá: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, xiv + 609 pp.
- Fernández, F. and S. Sendoya. 2004. Lista de las hormigas neotropicales. Biota Colombiana Volume 5, Number 1.
- Franco W., N. Ladino, J. H. C. Delabie, A. Dejean, J. Orivel, M. Fichaux, S. Groc, M. Leponce, and R. M. Feitosa. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674(5): 509-543.
- Kempf, W.W. 1972. Catalago abreviado das formigas da regiao Neotropical (Hym. Formicidae) Studia Entomologica 15(1-4).
- Lattke J. E. 2011. Revision of the New World species of the genus Leptogenys Roger (Insecta: Hymenoptera: Formicidae: Ponerinae). Arthropod Systematics and Phylogeny 69: 127-264
- Wheeler W. M. 1923. The occurrence of winged females in the ant genus Leptogenys Roger, with descriptions of new species. American Museum Novitates 90: 1-16.

