Mycocepurus castrator
| Mycocepurus castrator | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Attini |
| Genus: | Mycocepurus |
| Species: | M. castrator |
| Binomial name | |
| Mycocepurus castrator Rabeling & Bacci, 2010 | |
Mycocepurus castrator is an obligate, workerless social parasite of Mycocepurus goeldii and is so far known only from Rio Claro, Sao Paulo State, Brazil. Mycocepurus castrator occurs sympatrically with Mycocepurus smithii and Mycocepurus obsoletus, but cannot be confounded with any other Mycocepurus species because of its multiple morphological adaptations for a parasitic lifestyle.
| At a Glance | • Workerless Inquiline |
Identification
Rabeling & Bacci (2010) - Mycocepurus castrator can be recognized by the following characteristics: (i) the long antennal scapes surpassing the posterior margin of the head by half their length; (ii) reduced, blade-like mandibles lacking dentition of masticatory margin; (iii) concave shape of first gastric tergite; (iv) smooth and shiny body sculpture with hexagonal microsculpture; (v) reduced palpal formula (3,2); (vi) females and males with 11 antennal segments; (vii) males lacking serrated ventral border of aedeagus; (viii) absence of clear fenestra from forewings of queens and males; (ix) worker caste presumably absent; (x) metapleural gland orifice enlarged in females, and potentially present in males. Mycocepurus castrator males and females look extremely similar to each other, and males are distinguished most easily from the females by the genitalia protruding from the tip of the metasoma and their darker brown colour (vs reddish brown in the queens).
Distribution
Known only from Rio Claro, Sao Paulo State, Brazil.
Latitudinal Distribution Pattern
Latitudinal Range: -14.9121° to -22.3955°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: Brazil (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
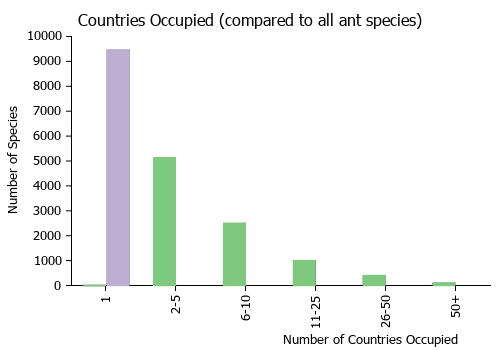
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
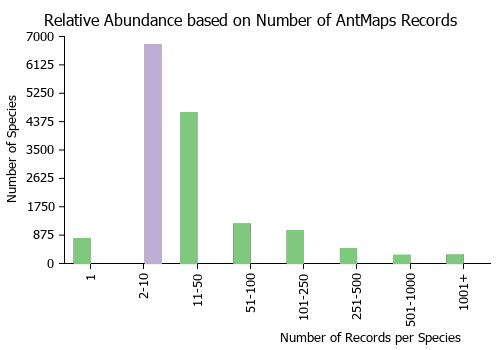
|
Biology
|
Mycocepurus castrator is an obligate, workerless social parasite of Mycocepurus goeldii. An investigation of its biology was undertaken and included in the publication that described the species (Rabeling and Bacci, 2010). All of the following details in this section are from this study.
Although the host ant is widespread and abundant throughout much of southern South America, M. castrator has been collected only twice, both times at the type locality (the Campus of Sao Paulo State University (UNESP) in Rio Claro, Brazil, 22.3955°S, 047.5424°W; elevation 608 m). . This suggests that parasite populations are probably few in number, small in size and patchily distributed. Observations of nesting biology and colony counts suggest that M. castrator is polygynous, host tolerant and allows for the production of sterile M. goeldii workers, whereas the production of host sexual offspring is suppressed in the presence of the parasite. The host, M. goeldii, appears to be monogynous in the Rio Claro population, but both mono- and polygynous colonies co-occur in the Brazilian Amazon.
Nesting Habits
Natural history and nest biology. Mycocepurus castrator has been found twice in adjacent nests of M. goeldii. The two host nests had five and eight chambers, respectively, which were distributed between 5 and 190 cm depth. The colony studied in 2006 contained 105 alate queens and 78 alate males of M. castrator, and 771 workers of M. goeldii. Dealate queens of either species could not be encountered, suggesting that the queenright chamber was either missed during the excavation or that the queens escaped into adjacent tunnels.
Colony Attributes
The 2008 colony contained 15 dealate and 66 alate M. castrator queens, only six alate males, 1034 M. goeldii workers, a single dealate M. goeldii queen and worker pupae. The parasite’s numerical male/female sex ratio was strongly female biased (6/66 = 0.09). Twelve of the 15 dealate M. castrator queens were encountered in the same fungus garden chamber as the reproductively active female of M. goeldii. Thus, M. castrator is host-queen tolerant. The other three dealate M. castrator queens were found together in a separate fungus chamber (chamber 1). The 12 queens encountered with the M. goeldii queen showed different reproductive activities: three were active egg layers, showing developed ovaries, yellow bodies and sperm-filled spermathecae. Thus, the parasite can be polygynous. In contrast, the remaining nine queens were prereproductive with filled spermathecae, but the ovaries were still developing, and yellow bodies were absent. The three dealate queens from chamber 1 were also prereproductive. The single M. goeldii queen was reproductively active.
The unparasitized M. goeldii colony studied in 2008 contained a single reproductively active queen, 33 alate queens, 496 workers and no males. During the excavation, males and queens were leaving the maternal colony for their nuptial flight, which started on 7 October.
A natural history study of M. goeldii in the Amazon Basin (Rabeling et al., 2007b) showed that some colonies had a single queen, whereas others were occupied by as many as four queens. Dissection of eight individuals from three separate colonies revealed that all of them were inseminated and had fully developed ovaries, demonstrating that these colonies were functionally polygynous.
Host species
Mycocepurus castrator has been found only in nests of M. goeldii and is so far only known from the type locality (Rio Claro, SP). Mycocepurus goeldii is a conspicuous, widely distributed species ranging approximately from the 40th to the 67th meridian west and from the 2nd to the 31st latitude south, an area covering most of Brazil, parts of Bolivia, Paraguay and northern Argentina. The range of habitats occupied by M. goeldii is remarkably diverse and ranges from Amazon rainforest, savannahs (Cerrado) to the fertile South American lowlands (Pampas), and secondary habitats disturbed by human activities. It does not occur in elevated sites of the South American Cordilleras. Mycocepurus goeldii workers can be distinguished clearly from its congeners based on the size and spine pattern of the mesosoma: it is the largest species in the genus and has the most complete set of spine pairs on the mesosoma. The natural history of this species has been studied near Sao Paulo City (Luederwaldt, 1918, 1926) and in the Manaus region of the Amazon Basin (Rabeling et al., 2007b), but these studies do not report the presence of a social parasite attacking M. goeldii. Like most inquilines for which we have data, M. castrator probably has a patchy and locally restricted distribution. In addition, it is probable that M. castrator is host specific, occurring only in nests of M. goeldii. Despite extensive excavation of nests of sympatrically occurring Mycocepurus species, the parasite was never encountered in the nests of M. smithii (Rabeling et al., 2009) or any other Mycocepurus species in Latin America (Rabeling, unpublished).
Reproduction
As soon as the uppermost chamber (CR081003-01) was opened during a nest excavation of a host colony, and the ants were transferred to the artificial nest chambers, M. castrator males started copulating with M. castrator females inside the artificial chamber (M. goeldii alates from an adjacent nest, placed into a laboratory nest, were never observed to copulate). During this time of ongoing mating activities, females and males ran erratically in jerky movements, and males mounted females seemingly at random. Observed copulations lasted between 18 and 27 s (n = 4). Single males attempted to copulate more than once. It is unknown whether repeated copulations resulted in successful transfer of sperm. Within 3 h after transfer to the artificial nest, three queens shed their wings, and subsequent dissections demonstrated that these females had been inseminated. However, their ovaries were still developing (i.e. ripe oocytes and yellow bodies were absent), indicating recent insemination (note: queens were preserved for dissection 2 weeks after copulation). Postcopulatory females did not tolerate mating attempts of males, and walked faster to outdistance their pursuer. After copulation, and wing shedding, the recently mated queens gathered and engaged in allogrooming, frequently licked each others meso- and metasomas, and wings for extensive periods of time. The first dead males were found 12 h after the mating event. Alates from other chambers did not copulate after transfer to the artificial nest. Potentially, the individuals in the topmost chamber were anticipating the upcoming nuptial flight and the opening of their nest chamber triggered the mating behaviour.
Behaviour
In the late afternoon of 29 September 2006, M. castrator was discovered when 31 queens and a single male left the host colony to aggregate on the nest mound. The dispersal activity was interrupted by rain, but continued on 2 October, when 24 queens and 72 males emerged. No further behavioural observations were made that year.
In 2008, M. goeldii colonies were excavated at the end of the dry season in order to study parasitized colonies before the nuptial flight. An approaching mating flight is easily identified in M. goeldii colonies, because the workers increase the number of nest entrances per soil mound to maximally 30 entrances, giving the nest mound a sponge-like appearance (Rabeling et al., 2009). Until 3 October, when a M. goeldii colony parasitized by M. castrator was encountered, the M. goeldii workers did not modify the nest mounds for mating flights. Upon excavating the parasitized colony, all individuals from a total of five nest chambers were transferred to artificial nest chambers for behavioural studies.
Host worker–parasite interactions. Host workers and parasite alates frequently antennated and interacted nonaggressively. Mycocepurus castrator alates did not require grooming by host workers because individuals cleaned themselves (i.e. licking appendages, cleaning antennae), and females groomed each other. Dealate M. castrator queens groomed M. goeldii workers, and were groomed by them also. On several occasions, M. goeldii workers licked the tip of a M. castrator metasoma for several minutes; it is not clear if the workers removed fecal droplets, or M. castrator queens laid either fertile or trophic eggs. Mycocepurus goeldii workers fed the parasite queens via trophallaxis. To be fed, M. castrator females frequently climbed onto the host workers’ backs, antennated the host’s antennae and head, until it bent its head backwards, regurgitated liquid, which was then consumed by the parasite. In addition to being fed, M. castrator males and females actively licked the fungus garden.
Three days after insemination, the host workers aggressively attacked one dealate queen from the topmost chamber (CR081003-01); six to eight workers secured her by the antennae, legs, head and petiole, until she died. Approximately 24 h after her death, three workers continued to carry around her corpse in the nest chamber. Six days after insemination, the host workers had attacked and killed several M. castrator queens, and had placed them on the refuse dump. Three dealate queens remained unmolested by hiding together in the fungus garden.
Host queen–parasite interactions. To observe the interactions between the host queen and the dealate M. castrator queens (n = 12), we placed the queens in a smaller nest chamber, after M. goeldii workers had arranged the fungus garden. The M. castrator queens were much more agile than the M. goeldii queen and initially walked around the nest chamber until they encountered a suitable spot; there they aggregated and started licking each other. When first placed in the chamber, the M. goeldii queen crawled under an adjacent piece of fungus garden and remained motionless; a worker then picked her up by the metasoma and moved the queen to a different position. During the carrying, the M. goeldii queen remained motionless. After several minutes, one M. castrator queen left the aggregation, ‘searching’ for the M. goeldii queen. When the host queen was encountered, she was surrounded by host workers antennating her. Regardless, the parasite climbed on the host queen’s back, and started licking her mesosoma, petiole, postpetiole and metasoma. Shortly afterwards, a second M. castrator queen joined the first; the M. goeldii queen continued to remain motionless. The remaining M. castrator queens eventually joined the grooming cluster, and alternated grooming themselves by pulling their legs and antennae through the tibio-tarsal cleaning apparatus of the foreleg, with grooming the host queen. When the first M. castrator queen climbed on the host queen’s back, the attending workers left and resumed fungus-gardening activities. They did not react aggressively to the parasite queens and often returned to antennate and to feed the host queen via trophallaxis. Once a worker carried the host queen to a different part of the fungus garden, and a M. castrator queen rode on her back during the location, licking her, and was not chased away. Either workers or M. castrator queens attended the host queen for most of time. Rarely and then for very short periods of time, she sat by herself. The M. castrator queens attempted constantly to climb on the backs of either other M. castrator queens, the host queen or host workers.
Introduction of parasite queen into a field colony. Two inseminated M. castrator queens from the topmost chamber (CR081003-01) were introduced to a M. goeldii colony, which opened its nest mound in preparation for the nuptial flight the previous day. The M. castrator queen was placed next to the nest mound. After orienting briefly, she immediately walked towards one of the entrance holes, and within a few seconds she disappeared into one of the entrances. The M. goeldii workers, which guarded the entrances, were not seen to attack, catch or struggle with the invading parasite. After 3 h the observation was stopped, and until then, M. goeldii workers had not expelled the M. castrator queen.
A second parasite queen was placed next to a M. goeldii colony, which had closed the supernumerary nest entrances after the nuptial flight. In contrast, the parasite did not start searching for the nest entrance and we repeatedly (five times) placed her on the side of the nest mound before she finally, perhaps by chance, walked over the nest entrance. When crossing the entrance, M. goeldii workers attacked the parasite immediately. We collected the parasite queen and a dissection identified her as recently inseminated with developing ovaries.
Introduction of parasite queen into a laboratory colony. To observe how M. castrator queens invade a M. goeldii colony, we maintained a nonparasitized M. goeldii colony in the laboratory. Upon transfer to the artificial nest, M. goeldii workers immediately covered the host queen with mycelial tufts, until she was completely hidden some 5 min later. To introduce the parasite queen, she was placed in a tube, which was connected to the fungus chamber. Quickly, she found her way out, headed directly towards the fungus garden, and immediately encountered the host queen. Then she started running in circles on the piece of fungus garden, under which the host queen was hidden. At that point, the host workers started chasing her, until one worker got hold of her petiole, and a second worker grabbed an antenna. The trio remained motionless for about 10 min, until both workers suddenly released the captive. Immediately, the M. castrator queen made a beeline for the host queen’s hiding place, where she was captured again. When being captured, the M. castrator queen assumed a characteristic position: she tucked her metasoma under the mesosoma, with the first gastric sternite touching the coxae. The combination of smooth body surface and broad, concave first gastric tergite provided little contact surface for attacking host workers, and their mandibles repeatedly slipped, upon which the host workers turned around and left. While being held by workers, the parasite queen was often antennated and licked at the base of the mandibles. After she was ‘dumped’ on the refuse pile, she promptly aimed for the host queen. This cycle of capture and release was repeated overnight. Meanwhile, the M. goeldii worker removed the fungus garden fragments from the host queen, releasing her from her mycelium prison. Unfortunately, the first contact of host and parasite queen was not observed, but early the next morning, the M. castrator queen was ‘riding’ on the M. goeldii queen, licking her mesosoma, petiole, postpetiole and metasoma. The M. goeldii workers frequently antennated and licked both queens, and the parasite queen was not attacked anymore. Subsequent observations revealed that the M. castrator queen was always ‘riding’ on top of the M. goeldii queen, and M. goeldii workers seemed to attend both queens equally.
Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- castrator. Mycocepurus castrator Rabeling & Bacci, 2010: 382, figs. 1A,C,E; 2A,C,E,G (q.m.) BRAZIL.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Queen
Holotype: HW 0.6, HL 0.64, SL 0.76, WL 1.07, PPW 0.62, PW 0.21, PL 0.24, PPL 0.19, CI 94, SI 127.
HW 0.6–0.65, HL 0.63–0.64, SL 0.73–0.8, WL 1.07–1.23, PPW 0.62–0.65, PW 0.21–0.25, PL 0.24–0.28, PPL 0.18–0.2, CI 94–104, SI 115–128 (n = 15).
Small species (WL 1.07) with a unique morphology reflecting the parasitic life history. In full face view, head rectangular (CI 94); sides approximately parallel, slightly tapering above mandibular insertions; head widest directly above the eyes; posterior margin of the head heart shaped, with a slight but distinct median concavity; posterolateral corners rounded, in lateral view drawn out to form a short, rounded lobe forming the ventrolateral corner of the head. Antennae with 11 segments; antennal scapes extremely long (SL 0.76), surpassing the posterior margin of the head by nearly half their length (SI 127). In full face view, frontal carinae and antennal scrobes absent. Frontal lobes small and rounded, barely covering the antennal sockets in frontal view. Median triangular portion of clypeus raised between the antennal insertions. Mandibles reduced, narrow, elongate, blade-like terminating in a pointed tooth; otherwise lacking teeth except for a small basal denticle. Maxillary palps reduced, with only three segments, labial palps with two segments. Ocelli slightly raised above the surface of vertex. Mesosoma with characteristic morphology related to wing bearing. Pronotal spines absent; propodeal spines well developed, stout, as wide as long at the base and sharply pointed; metapleural gland orifice very large and circular in oblique view, ventral margin forming small, vertical tooth. Petiole with a short peduncle; node triangular in side view, with sharp crest terminating in two thick pointed teeth. In dorsal view, postpetiole approximately 3× as wide as long (PPL 0.19, PPW 0.62); lateral borders tapering into pointed angles; translucent area near posterior margin forming broad u-shaped invagination. First gastric tergite strikingly concave in lateral view. Entire body surface more or less smooth and shiny, in most areas with hexagonal microsculpture resembling a honeycomb. Body sparsely covered with stiff setae; setae erect on vertex and frontal lobes, sub-decumbent on mesoscutum and scutellum, and appressed on postpetiole and metasoma. Wings infuscated with reduced venation, densely covered with setae; clear spot or fenestra in apical part of forewing absent; rsf1 faint, hardly visible. Colour: light to dark reddish-brown. – Paratype queens.
Male
HW 0.58–0.6, HL 0.58–0.6, SL 0.73–0.75, WL 1.1–1.2, PPW 0.63–0.65, PW 0.23–0.3, PL 0.25–0.28, PPL 0.18–0.2, CI 96–104, SI 121–126 (n = 15).
Paratypes. Remarkably similar to female, not resembling any other Mycocepurus male; characters as in female diagnosis with the following exceptions: head size of males smaller (HL 0.58–0.6, HW 0.58–0.6), whereas body length similar (WL 1.1–1.2). Mandibles reduced, narrow, elongate, blade-like, which do not terminate in a pointed tooth; otherwise lacking any teeth or denticles. Number of antennal segments reduced to 11; funicular segments approximately as long as broad, slowly increasing in length towards apex to 1.5× their width, only apical segment 5× as long as wide. Mesosoma lower and narrower; tiny opening present at the metapleuron, corresponding to the position of a metapleural gland opening in the female. First gastric tergite flat to slightly concave; male genitalia projecting forward from tip of metasoma. Basal apodeme lobed, separated from aedeagus by a deep constriction; ventral border lacking serration. Wing colour: medium to dark brown.
Type Material
Holotype, queen, BRAZIL: Sao Paulo, Rio Claro, Campus of Sao Paulo State University (UNESP), 22.3955°S, 047.5424°W, elevation 608 m, 29.ix.2006, C. Rabeling acc. no. CR060929-14, ex Mycocepurus goeldii nest. Holotype deposited at Museu de Zoologia da Universidade de Sao Paulo. Paratypes, 104 queens, 78 males, BRAZIL: same nest as holotype, 29.ix.2006–02.x.2006, col. C. Rabeling. Paratypes deposited at: American Museum of Natural History, BMEL, CRC, Museum of Comparative Zoology, MZSP, University of California, Davis, National Museum of Natural History.
Etymology
During collections of M. castrator, the host colonies were not observed to produce any alate queens and males, although sympatrically nesting M. goeldii colonies released alates. Therefore, we assume that the inquiline inhibits the host queens’ production of sexual offspring, allowing only for the production of the sterile worker caste. This is essentially ‘social castration’, hence the specific name ‘castrator’.
References
- Adams, R.M.M., Wells, R.L., Yanoviak, S.P., Frost, C.J., Fox, E.G.P. 2020. Interspecific Eavesdropping on Ant Chemical Communication. Frontiers in Ecology and Evolution 8. (doi:10.3389/fevo.2020.00024).
- Barros, L.A.C., Rabeling, C., Teixeira, G.A., dos Santos Ferreira Mariano, C., Delabie, J. H. C., de Aguiar, H. J. A. C. 2022. Decay of homologous chromosome pairs and discovery of males in the thelytokous fungus-growing ant Mycocepurus smithii. Scientific Reports 12, 4860 (doi:10.1038/s41598-022-08537-x).
- Cantone S. 2017. Winged Ants, The Male, Dichotomous key to genera of winged male ants in the World, Behavioral ecology of mating flight (self-published).
- de la Mora, A., Sankovitz, M., Purcell, J. 2020. Ants (Hymenoptera: Formicidae) as host and intruder: recent advances and future directions in the study of exploitative strategies. Myrmecological News 30: 53-71 (doi:10.25849/MYRMECOL.NEWS_030:053).
- Degueldre, F., Mardulyn, P., Kuhn, A., Pinel, A., Karaman, C., Lebas, C., Schifani, E., Bračko, G., Wagner, H.C., Kiran, K., Borowiec, L., Passera, L., Abril, S., Espadaler, X., Aron, S. 2021. Evolutionary history of inquiline social parasitism in Plagiolepis ants. Molecular Phylogenetics and Evolution 155, 107016 (doi:10.1016/j.ympev.2020.107016).
- Jesovnik, A., Sosa-Calvo, J., Lloyd, M.W., Branstetter, M.G., Andez. F.F., Schultz, T.R. 2017. Phylogenomic species delimitation and host-symbiont coevolution in the fungus-farming ant genus Sericomyrmex Mayr (Hymenoptera: Formicidae): ultraconserved elements (UCEs) resolve a recent radiation. Systematic Entomology 42, 523–542 (doi:10.1111/syen.12228).
- Rabeling, C. 2020. Social Parasitism. In: Starr, C. (ed.) Encyclopedia of Social Insects. Springer, Cham. (doi:10.1007/978-3-319-90306-4_175-1).
- Rabeling, C. & Bacci M. 2010. A new workerless inquiline in the Lower Attini (Hymenoptera: Formicidae), with a discussion of social parasitism in fungus-growing ants. Systematic Entomology 35:379-392.
- Rabeling, C., Messer, S., Lacau, S., do Nascimento, I.C., Bacci, M., Delabie, J.H.C. 2019. Acromyrmex fowleri: a new inquiline social parasite species of leaf-cutting ants from South America, with a discussion of social parasite biogeography in the Neotropical region. Insectes Sociaux. 66:435–451. doi:10.1007/s00040-019-00705-z
- Rabeling, C., Schultz, T.R., Pierce, N.E., Bacci, M. 2014. A social parasite evolved reproductive isolation from its fungus-growing ant host in sympatry. Current Biology 24, 2047–2052 (doi:10.1016/J.CUB.2014.07.048).
References based on Global Ant Biodiversity Informatics
- Rabeling C., S. Messer, S. Lacau, I. C. do Nascimento, M. Bacci Jr., and J. H. C. Delabie. 2019. Acromyrmex fowleri: a new inquiline social parasite species of leaf‑cutting ants from South America, with a discussion of social parasite biogeography in the Neotropical region. Insectes Sociaux https://doi.org/10.1007/s00040-019-00705-z
- Rabeling C., and M. Bacci. 2010. A new workerless inquiline in the Lower Attini (Hymenoptera: Formicidae), with a discussion of social parasitism in fungus-growing ants. Systematic Entomology 35: 379-392.


