AntWiki: The Ants --- Online
Nothing is known about the biology of Octostruma ascrobicula.
Identification
Longino (2013) - Antennal scrobe very shallow, not distinctly margined; face with arcuate carina; frontal carinae extending back and joining termini of facial arc (in Octostruma convallis, Octostruma convallisur, and Octostruma limbifrons, frontal carinae and facial arc do not join, the termini of the facial arc extend laterally beyond the termini of the frontal carinae); facial arc weak, semicircular, frontal carinae short, joining facial arc at level of compound eye (facial arc strong, less semicircular, frontal carinae longer, joining facial arc posterior to compound eye in Octostruma ascrobis); mesonotal foveolation extending onto dorsal pronotum, weakening anteriorly (mesonotum foveolate, pronotum contrastingly smooth and shining with sparse puncta in O. ascrobis); HW 0.42 (0.46–0.51 in O. ascrobis).
Keys including this Species
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: -0.199672° to -0.363°.
Neotropical Region: Ecuador (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species.
|
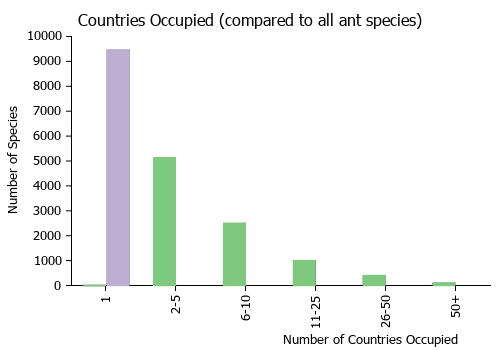
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species.
|
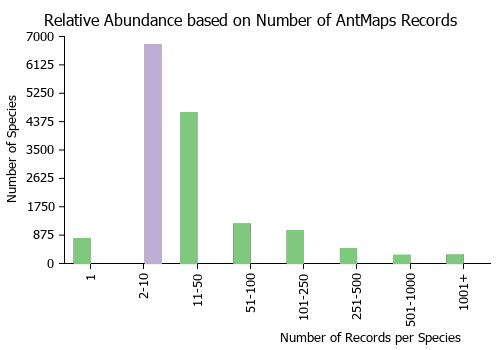
|
Biology
 Explore Overview of Octostruma biology Explore Overview of Octostruma biology
|
| Longino (2013) - Brown and Kempf (1960) summarized the biology of basicerotines as follows: The basicerotines all come from tropical or subtropical areas, and predominantly from mesic habitats, particularly rain forest, where they live primarily in the upper layers of the soil and in the soil cover, including large and small pieces of rotten wood. They are fairly common in soil cover berlesates. Nests have been found in snail shells, and in the peaty masses gathered about epiphytic ferns above the ground level. So far as is known, colonies are small, consisting of one or more dealate—or rarely ergatoid—females, and a few workers. Judging from the structure of the workers and females, one would suppose that they were predaceous on small arthropods...
Besides this summary, the behavior of three basicerotine species has been studied. Wilson (1956) observed a small captive colony of Eurhopalothrix biroi, a New Guinea species. Workers moved slowly and captured a variety of small, soft-bodied prey, including spiders, symphylans, entomobryid Collembola, campodeids, and hemipteran nymphs. Wilson and Brown (1984) observed a captive colony of Eurhopalothrix heliscata, a species from Singapore. The colony contained over 400 workers, multiple alate and dealate queens, several adult males, and brood. Foraging workers acted "rather like miniature ferrets," readily wedging themselves into small crevices. They foraged solitarily, attacking a variety of prey but mostly termites. They used their sharply-toothed mandibles to abruptly snap onto appendages of prey, maintaining purchase and slowly reaching around with the gaster to sting the prey. The strongly sclerotized labrum was also employed to press against the clamped appendage. The behavioral repertoire was limited. There did not appear to be trophallaxis, as workers and larvae fed directly from prey in the brood chambers. Nor did there appear to be any form of alarm communication. While there was generally an increase in the number of foragers when clusters of prey were presented, there was no evidence of any pheromone-based recruitment. Workers were non-aggressive and responded to disturbance by tucking the appendages and becoming immobile, often for minutes at a time. Wilson and Hölldobler (1986) studied captive colonies of Basiceros manni from Costa Rica and observed behavior not substantially different from E. heliscata. Foraging workers of many basicerotines are often encrusted with a firmly bonded layer of soil, which is thought to function as camouflage, enhancing crypsis (Hölldobler & Wilson, 1986).
Knowledge of the basic natural history of these ants has hardly progressed since the observations of Wilson, Brown, and Hölldobler. More specimens are now available for examination due to quantitative litter sampling, enhancing knowledge of basicerotine diversity and distribution, but discovering nests remains exceedingly difficult. Quantitative samples of 1 m2 litter plots reveals that small basicerotines can be very frequent, occurring in over 50% of samples in some cases, but never in large numbers. Individual samples usually contain fewer than ten workers, and workers are often accompanied by dealate queens. These results suggest that colonies, at least among New World species, are usually small, with tens of workers.
Less than half of the species of Octostruma have their queens described. Ergatoid queens are known from some species. Males are known from collections for some species but none have been described. The mating biology of these ants and how common ergatoid queens are across the genus and within colonies is not known.
|
|
Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- ascrobicula. Octostruma ascrobicula Longino, 2013: 17, figs. 6F, 16, 45 (w.) ECUADOR.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
HW 0.42, HL 0.41, WL 0.42, CI 103 (n=1). Differing from Octostruma ascrobis in the characters of the Diagnosis (see the identification section of this webpage); similar to O. ascrobis in most other respects (see complete description of A. ascrobis).
Type Material
Holotype worker: ECUADOR, Manabí: 78 km NE Chone [-0.363 -79.739 ±10km], 450 m, 9 Jun 1976, forest, ex sifted leaf litter (S. & J. Peck, B345) Museum of Comparative Zoology, unique specimen identifier MCZ-ENT00511413.
Etymology
The name refers to being a small version of Octostruma ascrobis. It is a noun in apposition and thus invariant.
References
- Longino, J.T. 2013. A revision of the ant genus Octostruma Forel 1912 (Hymenoptera, Formicidae). Zootaxa 3699, 1-61. doi:10.11646/zootaxa.3699.1.1





