Pogonomyrmex anergismus
| Pogonomyrmex anergismus | |
|---|---|

| |
| Conservation status | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Pogonomyrmecini |
| Genus: | Pogonomyrmex |
| Species group: | barbatus |
| Species: | P. anergismus |
| Binomial name | |
| Pogonomyrmex anergismus Cole, 1954 | |
These ants are workerless social parasites in nests of Pogonomyrmex barbatus and Pogonomyrmex rugosus. This species occurs at very low densities, infecting only a few nests out of a population of several hundred.
| At a Glance | • Workerless Inquiline |
Identification
Several highly distinctive characters in both castes make Pogonomyrmex anergismus easily identifiable. The male is unique among all known Pogonomyrmex s. s. males in that its pilosity is not flexuous and silky but erect and still. Superficially the sexes of anergismus are remarkably alike. Moreover, morphologic variation within the series of each sex is amazingly slight. This is well portrayed, in part, by the total lack of deviation in some of the measurements prefacing the descriptions of the sexes. (Cole 1968)
Distribution
Arizona, New Mexico and Texas.
Latitudinal Distribution Pattern
Latitudinal Range: 35.25055556° to 31.877205°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Nearctic Region: United States (type locality).
Neotropical Region: Mexico.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
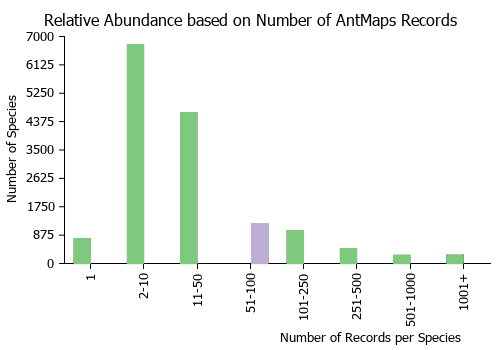
|
Habitat
Chihuahuan Desert grasslands.
Biology
The type nest, marked by a large, flat, gravel disc, contained large numbers of Pogonomyrmex rugosus workers and an estimated 200 males and alate females of Pogonomyrmex anergismus. Nearly as fast as the latter were exposed, rugosus workers carried them back into the nest galleries. There were no sexual forms of rugosus in the nest (Cole 1968).
Mackay and Mackay (2002) - These ants are social parasite in nests of Pogonomyrmex barbatus and Pogonomyrmex rugosus. This species occurs at very low densities, infecting only a few nests out of a population of several hundred. This species can be collected most efficiently by digging into nests of the host (to a depth of a few cms) in late summer or fall, and rapidly spreading the soil on the surface. After a quick examination, you should move on the next nest (you should notice them immediately if they are present, and to avoid stings by the host species). They are easily distinguished from the darker Pogonomyrmex rugosus by their light yellowish red color. They are essentially the same color as the alates of P barbatus, making them difficult to distinguish, but can be separated on the basis of their smaller size.
Flights occur during the afternoon after late summer and fall rains. Pouring about 8 liters of water on a parasitized colony can sometimes stimulate flights. Workers of the host are highly aggressive during the flights, as would happen during flights of their own species.
Females mate with colony mates, usually within the nest. The females fly from the nests, males remain in the nest. The spermathecae contain about 8000 sperm, about 1/20 that of other species of Pogonomyrmex (i. e. the other social parasite Pogonomyrmex colei). Apparently females must replenish the sperm in the spermatheca during later mating flights.
Apparently the females encounter aggression when they enter a nest of the host. When the female makes contact with the host queen or brood of the host, hostile behavior is replaced by intensive grooming by the host workers. Thereafter she is completely accepted by the nest and does not kill the host queen. Nests may contain more than one parasitic queen.
Genetics
The genome of P. anergismus was sequenced for a study examining how this and other parasitic ants with no worker caste may have altered their genome to arrive at a workerless state. In comparison to ants with a full complement of castes, there appeared to be no loss of genes in the parasitic ants. This suggests regulatory differences and not sequence differences predominate in gains and losses of castes (phenotypes). (Smith et al. 2015)
Castes
A workerless social parasite.
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- anergismus. Pogonomyrmex (Pogonomyrmex) anergismus Cole, 1954b: 115 (q.m.) U.S.A. See also: Cole, 1968: 46; Johnson, R.A. 1995: 257.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Queen
Cole (1968) - HL 1.52-1.56 mm, HW 1.67-1.71 mm, CI 109.6-109.9, SL 1.18-1.22 mm, SI 70.7-71.3, EL 0.38-0.38 mm, EW 0.27-0.27 mm, OI 24.4-25.0, WL 2.20-2.28 mm, PNL 0.38-0.38 mm, PNW 0.38-0.38 mm, PPL 0.46-0.46 mm, PPW 0.72-0.72 mm.
Mandible with 6 teeth arranged along a strongly oblique masticatory margin; subapical tooth about one-half length or apical, tightly appressed to it; first and second basals subequal in length, well separated from each other; penultimate basal tooth a little shorter than first and second basals, subequal in length to the broader ultimate basal which makes a straight angle with the basal mandibular margin; all teeth except ultimate basal moderately sharp.
Basal enlargement of antennal scape well flared, declivity or superior lobe meeting the shart at a broadly rounded angle; basal flange thin, extending to apex of superior lobe; lip weak, only slightly curved distad; point weak; longitudinal peripheral carina distinct.
Entire head with fine longitudinal rugulae which tend to fade out on occipital corners, the rugulae somewhat coarser just above mandibular articulations and on postgenae; interrugular spaces very faintly and finely punctulate; occipital corners with a few, shallow, elongate foveae.
Contours, in lateral view, of thorax, petiole, and postpetiole as portrayed in Pl. IX, Fig. 3; epinotal spines short, stubby, broad at the base, rather blunt; petiolar node in the shape of a weakly truncated cone, length of an terior and posterior declivities subequal, dorsum flattened; ventral process of petiolar peduncle weak to strong and subtriangular; ventral process of postpetiole weak. Conformation of petiole and postpetiole, viewed from above, as shown in Pl. XII, Fig. 14; petiolar node rather square, lateral margins subparallel, midregion with a distinct, broad, shallow impression, apex and apical corners truncate; postpetiole subglobular.
Pronotum with moderately fine transverse fugae; scutum finely, longitudinally rugulose; scutellum smooth, highly polished; sides of thorax with fine, longitudinal rugae; base of epinotum with coarser, subparallel, transverse rugae; declivious surface of epinotum smooth. Lateral and posterior surfaces of petiolar node with a few, fine, transverse rugulae or striae. Dorsum of postpetiole with a few, fine, irregular, transverse rugulae or striae. Interrugal spaces of thorax, petiole, and postpetiole free of sculpture. Gaster smooth without shagreening. Entire body shining.
Body with rather abundant, moderately long, mostly stiff and coarse, golden hairs; those on clypeus long and pointed as are also those comprising the well-developed psammophore; most of those elsewhere on head blunt, shortest on frontal region, longest on vertex and occipital border where they are largely clavate; sparse around compound eyes and in area between median ocellus and clypeus; long, fine, reclinate, and subappressed on antennal scapes, suberect on pedicel and first four flagellar segments, mostly subappressed and appressed on remaining segments. Hairs on thorax of variable length; longer and more pointed on pronotum; shorter and more clavate on scutum and scutellum; very short, blunt, and clavate on sides of thorax; long, slender, and pointed on fore coxae; very sparse and short on epinotum and on ventral process of petiolar peduncle; short and blunt on petiolar node; long and blunt on postpetiole. Hairs on gastric dorsum rather long, blunt, and evenly distributed.
Body color a rather uniform light ferrugineous red.
Male
Cole (1968) - HL 1.18-1.22 mm, HW 1.41-1.42 mm, CI 116.4-119.5, SL 0.80-0.84 mm, SI 56.7-59.2, EL 0.42-0.42 mm, EW 0.42-0.42 mm, OI 24.6-25.4, WL 2.01-2.05 mm, PNL 0.42-0.47 mm, PNW 0.42-0.47 mm, PPL 0.42-0.47 mm, PPW 0.76-0.76 mm.
Closely resembling the female in size, color, and general habitus. Conformation of mandible as shown in Pl. VIII, Fig. 15; blade narrow, apical margin straight for about two-thirds its length from the base, then curved evenly dorsad to meet the long, broad, apical tooth; masticatory margin subparallel with basal portion of apical margin, bearing 2 very broad, irregular, truncate teeth followed proximad by 2 much smaller, irregular teeth, then meeting imperceptibly the nearly straight, short, basal margin which is constricted proximally. Base of antennal scape trumpet-shaped. Cephalic rugulae absent except within and above ocellar triangle, beneath and immediately around compound eyes, and on postgenae.
Conformation, in lateral view, of thorax, petiole, and postpetiole as illustrated in Pl. IX, Fig. 2; epinotal spines angulate, short, very broad basally, blunt; apex of petiolar node less acute than that of the female; venter of petiolar peduncle with a prominent, broadly triangular process; ventral process of postpetiole moderately strong. Conformation of petiolar and postpetiolar nodes, viewed from above, as shown in Pl. XII. Fig. 13; petiolar node with a very broad and very distinct impression, apex evenly rounded, sides subparallel. Thoracic sculpture notably finer and sparser than that of the female, sometimes absent from dorsum.
In general, body hairs more abundant, more pointed, and more slender than in the female; rather evenly distributed; not flexuous; some have a tendency to curve near the tip; shorter than those of other Pogonomyrmex males; not silky; cephalic hairs long, slender, pointed, abundant, longest on ventral and occipital surfaces, mandibles, and clypeal border; hairs on petiole and postpetiole more robust than those on head and thorax; dorsum of petiolar peduncle without hairs or pubescence; ventral process of petiolar peduncle with a few rather long, straight, ventrally directed hairs; hairs on gaster long, slender, abundant, rather uniformly distributed, of approximately equal length. Gaster highly polished, without shagreening.
Head, thoracic dorsum, legs (except coxae), petiole, postpetiole, and gaster very shining; coxae and lateral portions of thorax glossy subopaque. Body color as in the female.
Type Material
Type locality: Fifteen miles east of Silver City, New Mexico, 6,900 feet. Cole Coll., Museum of Comparative Zoology, National Museum of Natural History, American Museum of Natural History, Creighton Coll., Gregg Coll.
References
- Mackay, W. P. and E. Mackay. 2002. The ants of New Mexico (Hymenoptera: Formicidae). Edwin Mellen Press, Lewiston, NY.
- Anderson, K. E.; Zeltzer, L.; Overson, R. P.; Clark, W. H. 2010. Identification of cryptic hosts for two inquiline parasites of the seed-harvester ant Pogonomyrmex and new localities for P. anergismus (Hymenoptera: Formicidae). The Southwestern Naturalist 55:532-538.
- Buschinger, A. (2009). Social parasitism among ants: a review. (Hymenoptera: Formicidae). Myrmecological News 12: 219-235.
- Cole, A. C., Jr. 1954. Studies of New Mexico ants. VII. The genus Pogonomyrmex with synonymy and a description of a new species (Hymenoptera: Formicidae). J. Tenn. Acad. Sci. 29: 115-121. (page 115, queen, male described)
- Cole, A. C., Jr. 1968. Pogonomyrmex harvester ants. A study of the genus in North America. Knoxville, Tenn.: University of Tennessee Press, x + 222 pp. (page 46, see also)
- Johnson, R.A. 1995. Distribution and natural history of the workerless inquiline ant Pogonomyrmex anergismus Cole. Psyche 101 (1994): 257-262. [1995.]
- Johnson, R.A. 2010. Independent colony founding by ergatoid queens in the ant genus Pogonomyrmex: queen foraging provides an alternative to dependent colony founding. Insectes Sociaux 57, 169–176 (doi:10.1007/s00040-010-0065-6).
- Mackay, W. P.; Van Vactor, S. 1985. New host record for the social parasite Pogonomyrmex anergismus (Hymenoptera: Formicidae). Proceedings of the Entomological Society of Washington 87:863. (page 863, host records)
- Parker, J.D., Rissing, S.W. 2002. Molecular evidence for the origin of workerless social parasites in the ant genus Pogonomyrmex. Evolution 56: 2017-2028.
- Smith, C. R., S. H. Cahan, C. Kemena, S. G. Brady, W. Yang, E. Bornberg-Bauer, T. Eriksson, J. Gadau, M. Helmkampf, D. Gotzek, M. O. Miyakawa, A. V. Suarez, and A. Mikheyev. 2015. How Do Genomes Create Novel Phenotypes? Insights from the Loss of the Worker Caste in Ant Social Parasites. Molecular Biology and Evolution. 32:2919-2931. doi:10.1093/molbev/msv165
References based on Global Ant Biodiversity Informatics
- Anderson K. E., L. Zeltzer, R. P. Overson, and W. H. Clark. 2010. Identification of cryptic hosts for two inquiline parasites of the seed-harvester ant Pogonomyrmex and new localities for P. anergismus (Hymenoptera: Formicidae). The Southwestern Naturalist 55: 532-538.
- Cover S. P., and R. A. Johnson. 20011. Checklist of Arizona Ants. Downloaded on January 7th at http://www.asu.edu/clas/sirgtools/AZants-2011%20updatev2.pdf
- Johnson R. Personnal Database. Accessed on February 5th 2014 at http://www.asu.edu/clas/sirgtools/resources.htm
- Johnson, R.A. 1994. Distribution and natural history of the workerless inquiline ant Pogonomyrmex anergismus Cole (Hymenoptera: Formicidae). Psyche 101:257-262.
- MacKay W. P., and S. Van Vactor. 1985. New host record for the social parasite Pogonomyrmex anergismus (Hymenoptera: Formicidae). Proceedings of the Entomological Society of Washington 87: 863.
- Mackay W. P., and E. E. Mackay. 2002. The ants of New Mexico (Hymenoptera: Formicidae). Lewiston, New York: Edwin Mellen Press, 400 pp.
- Parker, J.D. and S.W. Rissing. 2002. Molecular Evidence for the Origin of Workerless Social Parasites in the Ant Genus Pogonomyrmex. Evolution 56(10):2017-2028
- Pages using DynamicPageList3 parser function
- IUCN Red List vulnerable species
- Ant Associate
- Host of Pogonomyrmex barbatus
- Host of Pogonomyrmex rugosus
- Workerless Inquiline
- Need species key
- North temperate
- North subtropical
- Species
- Extant species
- Formicidae
- Myrmicinae
- Pogonomyrmecini
- Pogonomyrmex
- Pogonomyrmex anergismus
- Myrmicinae species
- Pogonomyrmecini species
- Pogonomyrmex species
- IUCN Red List
- Ssr


