Strumigenys microthrix
| Strumigenys microthrix | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Attini |
| Genus: | Strumigenys |
| Species: | S. microthrix |
| Binomial name | |
| Strumigenys microthrix (Kempf, 1975) | |
Occurs in mature wet forest habitats. Workers are found in leaf litter on the forest floor. Most collections are from Winkler samples of sifted leaf litter, but one collection is of two workers in a rotten stick in the leaf litter.
Identification
Bolton (2000) - A member of the Strumigenys schulzi-group. Together with Strumigenys cassicuspis and Strumigenys stauroma, microthrix forms a complex of closely related species, mainly known from Costa Rica. Most of the characters described above apply to all three species, but the pilosity of microthrix, or rather its lack of pilosity, distinguishes it from the other two, as follows.
S. microthrix: With head in occipital view the arc of the dorsum without projecting erect hairs. With head in profile dorsum of clypeus, and cephalic dorsum behind clypeus, without dense erect stubbly pilosity. With head in full-face view lateral margins of occipital lobes with minute anteriorly curved decumbent to appressed hairs.
S. cassicuspis and stauroma: With head in occipital view the arc of the dorsum with dense stubbly projecting erect hairs. With head in profile dorsum of clypeus, and cephalic dorsum behind clypeus, with dense erect short stubbly pilosity. With head in full-face view lateral margins of occipital lobes with numerous freely laterally projecting short erect stubbly hairs.
Apart from the size variation indicated above microthrix shows variation in colour from almost blackish brown to pale brownish yellow. Its petiole node in dorsal view is usually distinctly longer than broad but in some individuals, usually larger darker ones, the node may be only about as broad as long.
Longino (Ants of Costa Rica) - Mandibles in side view straight, not broadly curved ventrally; mandibles relatively short, subtriangular, much of the apical portion meeting along a serially toothed masticatory margin when closed (former Smithistruma); head relatively broad, CI 77-83; color red-brown to black; leading edge of the scape with entirely anteriorly directed short spatulate hairs; face punctate; sides of posterior half of mesosoma completely and densely punctulate; disc of postpetiole reticulate-punctate; ventral petiolar appendages entirely obsolete or present as very fine cariniform vestiges; infradental lamellae of propodeum vestigial or narrow and concave; gaster with 0-4 filiform (not spoon-shaped) erect setae; face lacking erect setae or with extremely inconspicous, short, subdecumbent setae. There is complex character variation (color from orange to dark gray, head length X width). Very similar to two other species: cassicuspis and stauroma. Both of these have a stubble of short setae covering the face.
Keys including this Species
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: 16.974° to 9.4817844°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: Colombia, Costa Rica (type locality), Honduras, Mexico, Nicaragua, Panama.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
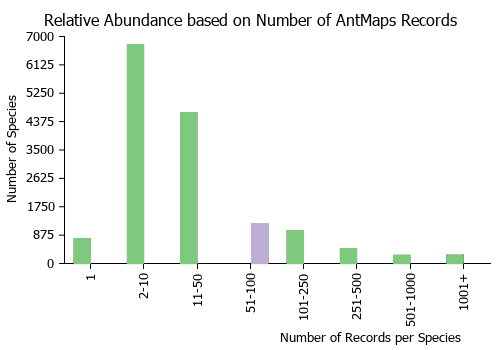
|
Biology
|
Castes
The INBIO collection contains a couple of queens of this species.
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- microthrix. Smithistruma microthrix Kempf, 1975a: 422, figs. 11, 12 (w.) COSTA RICA. Combination in Pyramica: Bolton, 1999: 1673; in Strumigenys: Baroni Urbani & De Andrade, 2007: 124. See also: Bolton, 2000: 222.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Bolton (2000) - TL 2.2-2.7, HL 0.64-0.80, HW 0.50-0.63, CI 77-83, ML 0.10-0.12, MI 14-16, SL 0.20-0.33, SI 50-55, PW 0.34-0.44, AL 0.66-0.78 (12 measured).
Anterior clypeal margin broadly convex. Dentition as described in introduction to group. Occipital lobes strongly expanded laterally in full-face view. Scape markedly dorsoventrally flattened and broad in dorsal view. Freely projecting hairs on leading edge of scape spoon-shaped , directed at right-angles to long axis of scape or curved toward the apex of the scape; no hairs conspicuously curved toward the scape base. Apicoscrobal and pronotal humeral hairs absent. With head in full-face view lateral margins of occipital lobes without stubbly projecting erect hairs; any hairs present are minute, curved forward and decumbent to appressed. Occipital lobes without minute peaks or tubercles. Dorsum of head without a transverse row of elongate standing hairs just behind highest point of vertex; clypeus and dorsum of head without erect short stubbly dense pilosity. Promesonotum in dorsal view flattened and very broad , without standing pilosity of any form. Femoral gland bullae elongate and conspicuous, especially on foreleg; bullae very obviously much longer than broad. Lateral spongiform lobes and posterior collar of petiole apparently absent, actually narrow and secondarily sclerotised, and pigmented so as to be virtually indistinguishable from the surrounding cuticle. With petiole in profile the dorsal length of the node distinctly greater than the height of its anterior face. Petiole in profile without trace of ventral spongiform tissue. Spongiform strip at base of first gastral sternite vestigial to absent. Entirety of head and alitrunk reticulate-punctate. First gastral tergite with very short basigastral costulae, behind which the sclerite is glassy smooth.
Type Material
Bolton (2000) - Holotype worker, COSTA RICA: Rio Toro Amarillo, vic. Guapiles, 26.ii.-9.iii.1966 (W. L. Brown) (Museum of Comparative Zoology) [examined].
References
- Baroni Urbani, C. & De Andrade, M.L. 2007. The ant tribe Dacetini: limits and constituent genera, with descriptions of new species. Annali del Museo Civico di Storia Naturale “G. Doria” 99:1-191.
- Bolton, B. 1999. Ant genera of the tribe Dacetonini (Hymenoptera: Formicidae). Journal of Natural History. 33:1639-1689. (page 1673, Combination in Pyramica)
- Bolton, B. 2000. The ant tribe Dacetini. Memoirs of the American Entomological Institute. 65:1-1028. (page 222, redescription of worker)
- Kempf, W. W. 1975a [1974]. Report on Neotropical Dacetine ant studies (Hymenoptera: Formicidae). Rev. Bras. Biol. 34: 411-424 (page 422, figs. 11, 12 worker described)
References based on Global Ant Biodiversity Informatics
- Bolton, B. 2000. The Ant Tribe Dacetini. Memoirs of the American Entomological Institute 65
- Brandao, C.R.F. 1991. Adendos ao catalogo abreviado das formigas da regiao neotropical (Hymenoptera: Formicidae). Rev. Bras. Entomol. 35: 319-412.
- Branstetter M. G. and L. Sáenz. 2012. Las hormigas (Hymenoptera: Formicidae) de Guatemala. Pp. 221-268 in: Cano E. B. and J. C. Schuster. (eds.) 2012. Biodiversidad de Guatemala. Volumen 2. Guatemala: Universidad del Valle de Guatemala, iv + 328 pp
- Dattilo W. et al. 2019. MEXICO ANTS: incidence and abundance along the Nearctic-Neotropical interface. Ecology https://doi.org/10.1002/ecy.2944
- Fernández, F. and S. Sendoya. 2004. Lista de las hormigas neotropicales. Biota Colombiana Volume 5, Number 1.
- Kempf W. W. 1975. Report on Neotropical Dacetine ant studies (Hymenoptera: Formicidae). Revista Brasileira de Biologia 34: 411-424.
- Longino J. T. 2013. Ants of Honduras. Consulted on 18 Jan 2013. https://sites.google.com/site/longinollama/reports/ants-of-honduras
- Longino J. T. L., and M. G. Branstetter. 2018. The truncated bell: an enigmatic but pervasive elevational diversity pattern in Middle American ants. Ecography 41: 1-12.
- Longino J. T., J. Coddington, and R. K. Colwell. 2002. The ant fauna of a tropical rain forest: estimating species richness three different ways. Ecology 83: 689-702.
- Longino J. T., and R. K. Colwell. 2011. Density compensation, species composition, and richness of ants on a neotropical elevational gradient. Ecosphere 2(3): 16pp.
- Longino J. et al. ADMAC project. Accessed on March 24th 2017 at https://sites.google.com/site/admacsite/
- Schonberg, L.A., J.T. Longino, N.M. Nadkarni and S.P. Yanoviak. 2004. Arboreal Ant Species Richness in Primary Forest, Secondary Forest, and Pasture Habitats of a Tropical Montane Landscape. Biotropica 36(3):402-409.

