Aphaenogaster floridana
| Aphaenogaster floridana | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Attini |
| Genus: | Aphaenogaster |
| Species: | A. floridana |
| Binomial name | |
| Aphaenogaster floridana Smith, M.R., 1941 | |
This species nests in sandy soil, with the nest being at least 80 cm deep. Nest entrances are often hidden with plant debris. Nests may be small, with fewer than 50 workers and apparently have a single queen. It is known to remove seeds (Atchison & Lucky, 2022).
Identification
The scape of the worker of this species extends about three funicular segments past the posterior lateral corner. The base of the scape is enlarged into a lobe. The dorsum of the pronotum is finely sculptured, but smooth and glossy, and the propodeum lacks spines (tiny angles may be present).
The scape other female is similar to that of the worker, surprisingly, the propodeum has well-developed spines. The pronotum is transversely striate, and only weakly shining.
It would be unlikely to confuse the workers of this species with any of the others, due to the unusual structure of the base of the scapes and the lack of propodeal spines.
Aphaenogaster floridana is one of the NA Aphaenogaster species without propodeal spines. The gaster is not significantly darker than the head and mesosoma (compared to Aphaenogaster boulderensis). They are found in sandy pine scrub and mixed hardwood forest. (DeMarco, 2015)
Florida
Deyrup (2016) - This is a yellow species with an arrowhead-shaped base on the antennal scape, but its most distinctive feature is the absence of propodeal spines, which are well developed in all other Florida species of Aphaenogaster.
Keys including this Species
Distribution
Florida
Deyrup (2016) - North Carolina south into Florida, west along the Gulf Coastal Plain into Alabama. In Florida, floridana is relatively common in uplands through north Florida, much rarer to the south, although it has been found on the Lake Wales Ridge in Highlands County.
Latitudinal Distribution Pattern
Latitudinal Range: 36.87157° to 27.135°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Nearctic Region: United States (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
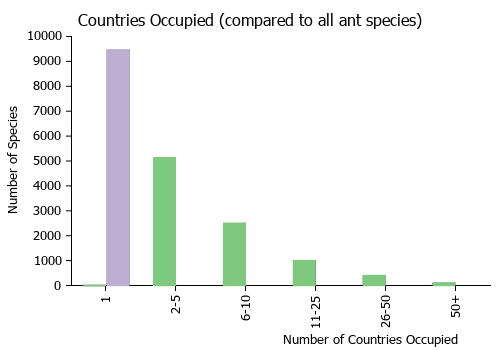
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Found in pine savannahs, longleaf pine, turkey oak forests, open shrubby habitats and sparse grassy habitats.
Florida
Deyrup (2016) - The following paragraph is summarized from Carroll (1975). Nests are in well-drained, sandy soil in open areas such as fields, sandhills, or Florida scrub habitats. There are usually one or two nest entrances, sometimes with a short turret of plant debris and arthropod remains, and often situated at the base of a clump of grass. The main entrance may be changed every few days or weeks. Nests are deeper than those of other Florida Aphaenogaster, sometimes 1.3 meters deep. Foraging is on the ground, crepuscular or nocturnal during warm weather. Foragers collect various arthropods, including Pheidole morrisii, and alate dispersing Solenopsis invicta and Solenopsis geminata. This species collects seeds of the hemiparasitic plant Seymeria pectinata, and pieces of Russula mushrooms. Alates are in the nest from late June and early July, and may be seen at nest entrances in late August and early September. The largest colony seen had approximately 200 workers.
In North Carolina, Carter (1962) found floridana on the Coastal Plain in grassy, open sandhill areas, and similar habitats, including grassy depressions between coastal dunes. In Florida, Van Pelt (1958) found floridana in open sandhill areas and open, sandy roadsides. He states that most foraging is nocturnal or on cloudy days.
Lack of Propodeal Spines
Deyrup (2016) - Propodeal spines are lacking in this species, while they are well developed in all other Florida species of Aphaenogaster. Males and queens have small spines, suggesting that the loss of spines in the worker is a recent evolutionary event. The adaptive significance of this is unknown, but two other Florida ants of open, sandy areas, Pogonomyrmex badius and Pheidole morrisii, also seem to have recently dispensed with propodeal spines, although they occasionally appear in P. badius. The primary function of the propodeal spines is to protect the vulnerable petiolar joint, best achieved by raising the petiole so that it rests between the petiolar spines. This is probably most useful for species that grapple and sting, and would not be as useful for species that spray repellents. Three possible hypotheses explaining the loss of propodeal spines in floridana are as follows: this species uses another form of defense, such as powerful chemical deterrents; this species has a way of avoiding grappling encounters; this species lives in a habitat where most competitive, aggressive, or defensive encounters are with other ants that spray repellent chemicals.
Castes
Queen
Images from AntWeb
   
| |
| Queen (alate/dealate). Specimen code casent0103584. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
Male
Images from AntWeb
    
| |
| Male (alate). Specimen code casent0103580. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ABS, Lake Placid, FL, USA. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- floridana. Aphaenogaster (Attomyrma) floridana Smith, M.R. 1941: 118 (w.) U.S.A. (Florida).
- Type-material: holotype worker, 1 paratype worker.
- Type-locality: holotype U.S.A.: Florida, Gretna, 15.vii.1906 (A.W. Morrill); paratype with same data.
- Type-depository: USNM.
- Mackay & Mackay, 2017: 248 (q.m.); Wheeler, G.C. & Wheeler, J. 1960b: 7 (l.).
- Status as species: Smith, M.R. 1944d: 14; Gregg, R.E. 1949b: 173; Creighton, 1950a: 143; Smith, M.R. 1951a: 796; Smith, M.R. 1958c: 117; Smith, M.R. 1967: 352; Smith, D.R. 1979: 1361; Deyrup, et al. 1989: 95; Bolton, 1995b: 69; Deyrup, 2003: 44; Coovert, 2005: 45; MacGown & Forster, 2005: 71; Deyrup, 2017: 47; Mackay & Mackay, 2017: 247 (redescription).
- Distribution: U.S.A.
Type Material
- Holotype, worker, Gretna, Florida, United States, 30°37′0″N 84°40′0″W / 30.616667°N 84.666667°W, 15 July 1906, A.W. Morrill, USNM No. 55660, National Museum of Natural History.
- Paratype, 1 worker, Gretna, Florida, United States, 30°37′0″N 84°40′0″W / 30.616667°N 84.666667°W, 15 July 1906, A.W. Morrill, USNM No. 55660, National Museum of Natural History.
Description
References
- Atchison, R. A., Lucky, A. 2022. Diversity and resilience of seed-removing ant species in Longleaf Sandhill to frequent fire. Diversity 14, 1012 (doi:10.3390/d14121012).
- Bolton, B. 1995b. A new general catalogue of the ants of the world. Cambridge, Mass.: Harvard University Press, 504 pp. (page 69, catalogue)
- DeMarco, B.B. 2015. Phylogeny of North American Aphaenogaster species (Hymenoptera: Formicidae) reconstructed with morphological and DNA data. Ph.D. thesis, Michigan State University.
- Deyrup, M.A. 2016. Ants of Florida: Identification and Natural History. CRC Press, 423 pp.
- Lau, M.K., Ellison, A.M., Nguyen, A., Penick, C., DeMarco, B., Gotelli, N.J., Sanders, N.J., Dunn, R.R., Helms Cahan, S. 2019. Draft Aphaenogaster genomes expand our view of ant genome size variation across climate gradients. PeerJ 7, e6447 (doi:10.7717/PEERJ.6447).
- Lau, M.K., Ellison, A.M., Nguyen, A., Penick, C., DeMarco, B., Gotelli, N.J., Sanders, N.J., Dunn, R.R., Helms Cahan, S. 2019. Draft Aphaenogaster genomes expand our view of ant genome size variation across climate gradients. PeerJ 7:e6447 (DOI 10.7717/peerj.6447).
- MacGown, J.A., Booher, D., Richter, H., Wetterer, J.K., Hill, J.G. 2021. An updated list of ants of Alabama (Hymenoptera: Formicidae) with new state records. Transactions of the American Entomological Society 147: 961-981 (doi:10.3157/061.147.0409).
- Perez-Delgado, A.J., Lugo, D., Santos-Perdomo, I., Jimenez-Garcia, E., Suarez, D. 2023. Rediscovery of the Canary Islands endemic Aphaenogaster hesperia Santschi, 1911 (Hymenoptera, Formicidae, Myrmicinae). Zootaxa 5383(1), 67–74 (doi:10.11646/zootaxa.5383.1.5).
- Smith, M. R. 1941. Two new species of Aphaenogaster (Hymenoptera: Formicidae). Great Basin Nat. 2: 118-121.(page 118, worker described)
- Tschinkel WR. 2011. The nest architecture of three species of north Florida Aphaenogaster ants. Journal of Insect Science 11:105.
- Tschinkel, W.R. 2015. The architecture of subterranean ant nests: beauty and mystery underfoot. Journal of Bioeconomics 17:271–291 (DOI 10.1007/s10818-015-9203-6).
- Wheeler, G. C.; Wheeler, J. 1960b. Supplementary studies on the larvae of the Myrmicinae (Hymenoptera: Formicidae). Proc. Entomol. Soc. Wash. 62: 1-32 (page 7, larva described)
References based on Global Ant Biodiversity Informatics
- Annotated Ant Species List Ordway-Swisher Biological Station. Downloaded at http://ordway-swisher.ufl.edu/species/os-hymenoptera.htm on 5th Oct 2010.
- Del Toro, I. 2010. PERSONAL COMMUNICATION. MUSEUM RECORDS COLLATED BY ISRAEL DEL TORO
- Deyrup M., C. Johnson, G. C. Wheeler, J. Wheeler. 1989. A preliminary list of the ants of Florida. Florida Entomologist 72: 91-101
- Deyrup M., L. Deyrup, and J. Carrel. 2013. Ant Species in the Diet of a Florida Population of Eastern Narrow-Mouthed Toads, Gastrophryne carolinensis. Southeastern Naturalist 12(2): 367-378.
- Deyrup, M. 2003. An updated list of Florida ants (Hymenoptera: Formicidae). Florida Entomologist 86(1):43-48.
- Deyrup, M. and J. Trager. 1986. Ants of the Archbold Biological Station, Highlands County, Florida (Hymenoptera: Formicidae). Florida Entomologist 69(1):206-228
- Graham J.H., H.H. Hughie, S. Jones, K. Wrinn, A.J. Krzysik, J.J. Duda, D.C. Freeman, J.M. Emlen, J.C. Zak, D.A. Kovacic, C. Chamberlin-Graham, H. Balbach. 2004. Habitat disturbance and the diversity and abundance of ants (Formicidae) in the Southeastern Fall-Line Sandhills. 15pp. Journal of Insect Science. 4: 30
- Graham, J.H., A.J. Krzysik, D.A. Kovacic, J.J. Duda, D.C. Freeman, J.M. Emlen, J.C. Zak, W.R. Long, M.P. Wallace, C. Chamberlin-Graham, J.P. Nutter and H.E. Balbach. 2008. Ant Community Composition across a Gradient of Disturbed Military Landscapes at Fort Benning, Georgia. Southeastern Naturalist 7(3):429-448
- Guénard B., K. A. Mccaffrey, A. Lucky, and R. R. Dunn. 2012. Ants of North Carolina: an updated list (Hymenoptera: Formicidae). Zootaxa 3552: 1-36.
- Ivanov K., L. Hightower, S. T. Dash, and J. B. Keiper. 2019. 150 years in the making: first comprehensive list of the ants (Hymenoptera: Formicidae) of Virginia, USA. Zootaxa 4554 (2): 532–560.
- Johnson C. 1986. A north Florida ant fauna (Hymenoptera: Formicidae). Insecta Mundi 1: 243-246
- Longino, J.T. 2010. Personal Communication. Longino Collection Database
- MacGown J. A., J. G. Hill, and M. Deyrup. 2009. Ants (Hymenoptera: Formicidae) of the Little Ohoopee River Dunes, Emanuel County, Georgia. J. Entomol. Sci. 44(3): 193-197.
- Trager, J. and C.Johnson. 1985. A slave-making ant in Florida: Polyergus lucidus with observations on the natural history of its host Formica archboldi (Hymenoptera: Formicidae). The Florida Entomologist 68(2):261-266.
- Van Pelt A. F. 1948. A Preliminary Key to the Worker Ants of Alachua County, Florida. The Florida Entomologist 30(4): 57-67
- Van Pelt A. F. 1966. Activity and density of old-field ants of the Savannah River Plant, South Carolina. Journal of the Elisha Mitchell Scientific Society 82: 35-43.
- Van Pelt A., and J. B. Gentry. 1985. The ants (Hymenoptera: Formicidae) of the Savannah River Plant, South Carolina. Dept. Energy, Savannah River Ecology Lab., Aiken, SC., Report SRO-NERP-14, 56 p.
- Whitcomb W. H., H. A. Denmark, A. P. Bhatkar, and G. L. Greene. 1972. Preliminary studies on the ants of Florida soybean fields. Florida Entomologist 55: 129-142.

