Harpagoxenus canadensis
| Harpagoxenus canadensis | |
|---|---|

| |
| Conservation status | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Myrmicinae |
| Tribe: | Crematogastrini |
| Genus: | Harpagoxenus |
| Species: | H. canadensis |
| Binomial name | |
| Harpagoxenus canadensis Smith, M.R., 1939 | |
This species is a social parasite. In the original description Smith reported the type material was accompanied by 24 workers of the host species Leptothorax muscorum (as Leptothorax acervorum subsp. canadensis). It has also been reported with Leptothorax canadensis (Beibl et al., 2005; de la Mora et al., 2021).
| At a Glance | • Dulotic • Tandem running |
Identification
Smith (1939) - The ergatoid female of Harpagoxenus canadensis can be distinguished from the worker of Temnothorax americanus by the narrower and deeper median emargination of the anterior border of the clypeus, the shallower antennal scrobes, the toothless or finely denticulate mandibles, and the blunter, superior border of the petiole. It differs from the ergatoid female of Harpagoxenus sublaevis in the following characters: Posterior border of head not so strongly emarginate; clypeus with a narrow median emargination on its anterior border; no erect hairs on the antennal scapes; antennal scrobes much shallower; petiole lacking the sharp lateral carinae; and postpetiole without the long, distinct, ventral tooth.
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: 53.941° to 39.052017°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Nearctic Region: Canada (type locality), United States.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
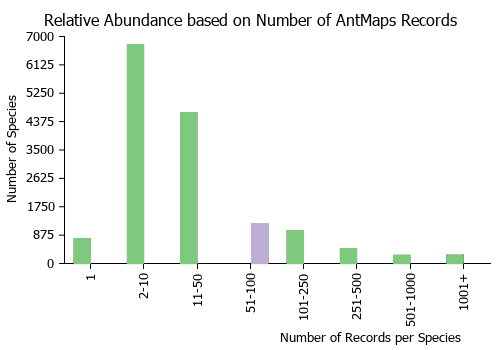
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
|
Biology
Life History Traits
- Queen number: monogynous (Rissing and Pollock, 1988; Frumhoff & Ward, 1992) (queenless worker reproduction)
- Queen type: winged (Rissing and Pollock, 1988; Frumhoff & Ward, 1992) (queenless and queen-right worker reproduction)
- Queen mating frequency: single (Rissing and Pollock, 1988; Frumhoff & Ward, 1992)
- Worker-produced males: present (Buschinger & Alloway, 1978;Frumhoff & Ward, 1992)
Castes
Worker
Images from AntWeb
   
| |
| Worker. Specimen code casent0006075. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by UCDC, Davis, CA, USA. |
Queen
Images from AntWeb
    
| |
| Queen (alate/dealate). Specimen code casent0178586. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by MCZ, Cambridge, MA, USA. |
Male
Images from AntWeb
   
| |
| Male (alate). Specimen code casent0006078. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by UCDC, Davis, CA, USA. |
     
| |
| Male (alate). Specimen code casent0178587. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by MCZ, Cambridge, MA, USA. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- canadensis. Harpagoxenus canadensis Smith, M.R. 1939b: 168, figs. 1A,B, 2A (q., ergatoid q.) CANADA (Québec).
- Type-material: 3 syntype queens, 9 syntype ergatoid queens.
- Type-locality: Canada: Québec Prov., Québec, from Polyporus on birch (J.I. Beaulne).
- Type-depositories: AMNH, CNCO, LACM, MCZC, USNM.
- Gregg, 1945a: 74 (w.).
- Status as species: Gregg, R.E. 1945a: 74; Creighton, 1950a: 284; Smith, M.R. 1951a: 822; Kutter, 1968b: 204; Francoeur, 1977b: 207; Smith, D.R. 1979: 1398; Buschinger, 1981: 213; Wheeler, G.C., et al. 1994: 302; Bolton, 1995b: 211; Coovert, 2005: 81; Ellison, et al. 2012: 243; Xu, 2012a: 21 (in key).
- Distribution: Canada, U.S.A.
Type Material
Quebec, Province of Quebec, Canada (Jos. I. Beaulne). Cotypes No. 53248, U.S. National Museum. Described from nine ergatoid females and three queens which were collected from a fungus, Polyporus sp., on birch. Eight ergatoid females and two queens have been deposited in the U. S. National Museum, two ergatoid females and one queen in the Canadian National Collections, Ottawa, Canada, and one ergatoid female each in the American Museum of Natural History and the Museum of Comparative Zoology (Harvard University). Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Queen
Ergatoid Length 4-4.25 mm.
Head subrectangular, distinctly longer than broad, with very feebly emarginate posterior border, rounded occipital angles, and moderately convex sides. Eye convex, separated from base of mandible by a distance greater than eye's widest diameter. Mandible rather small, less convex than that of americanus, masticatory border obliquely, concavely curved, toothless or very finely denticulate. Anterior border of clypeus with a rather deep median emargination, which is narrow (much narrower than that of americanus); posterior border of clypeus broadly rounded, extending backward between frontal carinae. Frontal area not clearly defined. Frontal carinae subparallel, not strongly elevated, extending beyond apices of antennal scapes but not forming such deep and distinct scrobes as in americanus. Antenna 11-segmented; scape stout, curved, and strongly depressed; last three segments of funiculus greatly enlarged, ultimate segment exceeding the combined length of the two preceding segments, funicular segments 2-6 each clearly broader than long. Vertex with a distinct median and two extremely small, indistinct lateral ocelli. Thorax, from above, with distinct promesonotal, and mesoepinotal sutures, the mesoepinotal region strongly constricted; epinotum with a pair of moderately long, coarse spines which are directed upward, backward, and outward. Petiole, viewed laterally, with convex posterior surface, which meets the anterior surface in such a manner as to form a bluntly angular node; ventral surface of peduncle with a short, blunt, anterior tooth, but no prominent midventral plate as in sublaevis; viewed posteriorly, sides of petiole converging toward superior border, which is narrow laterally and faintly emarginate. Postpetiole distinctly broader than long, broader anteriorly than posteriorly, and with pronounced angular humeri; lacking ventrally the prominent tooth or spine of sublaevis, but with a very weak anterior tubercle. Base of gaster much constricted, very little broader than the postpetiole, and with slightly angular humeri.
Mandibles, anterior surface of petiole, and gaster mostly smooth and shining, remainder of body subopaque; front of head with longitudinal striae, remainder of head, excepting clypeus, posterior border of head, and occipital angles, reticulate punctate, the cheeks, however, with longitudinal rugulae in addition; thorax, petiole, and postpetiole varying from reticulate punctulate to rugulose reticulate.
Hairs moderately long, suberect to erect, sparsely distributed over body and on coxae, trochanters, and femora, especially the ventral surfaces of the two latter. Pubescence rather long and coarse but sparse, closely appressed on the body, suberect on the appendages.
Head, gaster, and masticatory border of mandibles deep brownish black; thorax, petiole, postpetiole, and appendages lighter brown.
Intermediate ergatoid. Length 4.10 mm.
Thorax, viewed from above, differing from that of the queen as follows: Paraptera of mesothorax fused with scutum; anterior wings represented by black, sclerotized stubs; scutellum not clearly separated from preceding part of mesonotum, trapezoidal, wider anteriorly than posteriorly; paraptera of metathorax fused with metanotum, forming a trapezoidal area similar to scutellum but smaller; posterior pair of wings represented on each side by a small rough area, out of which projects a whitish appendage; epinotum like that of queen; petiole, viewed laterally, with posterior surface less convex than in the simple ergatoid female, and therefore more acutely angular. Sculpturing very similar to that of the simple ergatoid female but with a rather distinct row of transverse foveolate punctures anterior to the scutellum. This form represents an intermediate stage between the simple ergatoid female (which is decidedly more workerlike) and the more structurally complex queen. The drawings representing a dorsal view of the thorax of each of the three forms (fig. 2, A, B, C) show the differences between them.
Length 4.5-5 mm.
Larger than the ergatoid female. Also differing in the structure of the thorax. Similar in other ways except for the following differences: Anterior angles of head more protuberant. Posterior border of head more deeply emarginate. Ocelli larger and more conspicuous. Posterior surface of petiole less convex, thus causing the dorsum of the node to appear more acutely angular, when the petiole is viewed in lateral profile. Ventral surface of peduncle with a slight median plate, which ends in a blunt tooth anteriorly. Thorax, from above, with subangular humeri. Scutum of mesothorax bluntly angular anteriorly, forming an angle of considerably less than 90 degrees; parapsidal furrows present. Sculpturing similar to that of the ergatoid female but posterior surface of head more shining. Scutum and scutellum with a few weak but distinct longitudinal rugulae.
Karyotype
- n = 18 (Canada) (Buschinger et al., 1980; Fischer, 1987).
Worker Morphology
 Explore: Show all Worker Morphology data or Search these data. See also a list of all data tables or learn how data is managed.
Explore: Show all Worker Morphology data or Search these data. See also a list of all data tables or learn how data is managed.
- Caste: monomorphic
References
- Baer, B. 2011. The copulation biology of ants (Hymenoptera: Formicidae). Myrmecological News 14: 55-68.
- Beibl, J., Stuart, R.J., Heinze, J., Foitzik, S. 2005. Six origins of slavery in formicoxenine ants. Insectes Sociaux 52, 291–297 (doi:10.1007/s00040-005-0808-y).
- Borowiec, M.L., Cover, S.P., Rabeling, C. 2021. The evolution of social parasitism in Formica ants revealed by a global phylogeny. Proceedings of the National Academy of Sciences 118, e2026029118 (doi:10.1073/pnas.2026029118).
- Buschinger, A. 2009. Social parasitism among ants: a review (Hymenoptera: Formicidae). Myrmecological News 12: 219-235.
- Buschinger, A., Alloway, T.M. (1979): Sexual behaviour in the slave-making ant, Harpagoxenus canadensis M.R. SMITH, and sexual pheromone experiments with H. canadensis, H. americanus (EMERY), and H. sublaevis (NYLANDER)(Hymenoptera; Formicidae). Z. Tierpsychol. 49, 113-119.
- Cantone S. 2018. Winged Ants, The queen. Dichotomous key to genera of winged female ants in the World. The Wings of Ants: morphological and systematic relationships (self-published).
- de la Mora, A., Sankovitz, M., Purcell, J. 2020. Ants (Hymenoptera: Formicidae) as host and intruder: recent advances and future directions in the study of exploitative strategies. Myrmecological News 30: 53-71 (doi:10.25849/MYRMECOL.NEWS_030:053).
- D'Ettorre, P., Heinze, J. 2001. Sociobiology of slave-making ants. Acta ethologica 3, 67–82 (doi:10.1007/s102110100038).
- Grasso, D.A., Visicchio, R., Castracani, C., Mori, A., Le Moli, F. 2003. The mandibular glands as a source of sexual pheromones in virgin queens of Polyergus rufescens (Hymenoptera, Formicidae). Italian Journal of Zoology 70, 229–232 (doi:10.1080/11250000309356522).
- Gregg, R. E. 1945b. The worker caste of Harpagoxenus canadensis Smith (Formicidae). Can. Entomol. 77: 74-76. (page 74, worker described)
- Heinze, J., Stewart, R.J., Alloway, T.M., Buschinger, A. (1992): Host specificity in the slave-making ant Harpagoxenus canadensis M.R. SMITH. Can. J. Zool. 70, 167-170.
- Ivanov, K. 2019. The ants of Ohio (Hymenoptera, Formicidae): an updated checklist. Journal of Hymenoptera Research 70: 65–87 (doi:10.3897@jhr.70.35207).
- Johnson, C.A. 2000. Mechanisms of dependent colony founding in the slave-making ant, Polyergus breviceps Emery (Hymenoptera: Formicidae). Ph.D. thesis, City University of New York.
- Smith, M. R. 1939b. The North American ants of the genus Harpagoxenus Forel, with the description of a new species (Hymenoptera: Formicidae). Proc. Entomol. Soc. Wash. 41: 165-172. (page 168, figs. 1, 2 queen described)
References based on Global Ant Biodiversity Informatics
- Francoeur A. 2001. Les fourmis de la forêt boréale (Formicidae, Hymenoptera). Le naturaliste canadien 125(3): 108-114.
- Glasier J. R. N., J. H. Acorn, S. E. Nielsen, and H. Proctor. 2013. Ants (Hymenoptera: Formicidae) of Alberta: A key to species based primarily on the worker caste. Canadian Journal of Arthropod Identification 22: 1-104.
- Glasier J. R. N., S. E. Nielsen, J. Acorn, and J. Pinzon. 2019. Boreal sand hills are areas of high diversity for Boreal ants (Hymenoptera: Formicidae). Diversity 11, 22; doi:10.3390/d11020022.
- Glasier J. R. N., S. Nielsen, J. H. Acorn, L. H. Borysenko, and T. Radtke. 2016. A checklist of ants (Hymenoptera: Formicidae) of Saskatchewan. The Canadian Field-Naturalist 130(1): 40-48.
- Gregg R. E. 1946. The ants of northeastern Minnesota. American Midland Naturalist 35: 747-755.
- Gregg, R.E. 1945. The worker caste of Harpagoxenus candadensis Smith (Formicidae). Canadian Entomologist 77(4):74-76
- Ivanov, K. 2019. The ants of Ohio (Hymenoptera, Formicidae): an updated checklist. Journal of Hymenoptera Research 70: 65–87.
- Smith M. R. 1939. The North American ants of the genus Harpagoxenus Forel, with the description of a new species (Hymenoptera: Formicidae). Proceedings of the Entomological Society of Washington 41: 165-172.
- Stuart, R.J. and T.M. Alloway. 1985. Behavioural evolution and domestic degeneration in obligatory slave-making ants (Hymenoptera: Formicidae: Leptothoracini). Animal Behavior 33:1080-1088
- Pages using DynamicPageList3 parser function
- IUCN Red List vulnerable species
- Ant Associate
- Host of Leptothorax muscorum
- Host of Leptothorax canadensis
- Dulotic
- Tandem running
- Need species key
- North temperate
- Karyotype
- Species
- Extant species
- Formicidae
- Myrmicinae
- Crematogastrini
- Harpagoxenus
- Harpagoxenus canadensis
- Myrmicinae species
- Crematogastrini species
- Harpagoxenus species
- Need Body Text
- IUCN Red List




