Prionopelta kraepelini
| Prionopelta kraepelini | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Amblyoponinae |
| Tribe: | Amblyoponini |
| Genus: | Prionopelta |
| Species: | P. kraepelini |
| Binomial name | |
| Prionopelta kraepelini Forel, 1905 | |
In some SE Asian areas, collected from leaf litter samples in forested areas (mainly rainforests but including parkland on volcanic soil). In other locales it is primarily found in disturbed sites. The contrasting habitats that it has been found in, along with collections occurring across a wide range, suggests this ant is a tramp species.
| At a Glance | • Larval Hemolymph Feeding • Limited invasive |
Identification
Shattuck (2008) - Sculpturing on dorsum of pronotum consisting of fine punctations which contrast markedly with widely spaced foveae on mesonotum and propodeum, the foveae on the propodeum varying in density across its width (weakest medially, stronger laterally). Head width less than 0.48mm. Petiole relatively narrow, PetW less than 0.21.
Brown (1960) confused this species with Prionopelta opaca and didn’t recognize the specimens here placed in Prionopelta robynmae as belonging to a separate species. In fact, all three of these species, while morphologically similar, can be separated as follows. In true kraepelini the sculpturing on the pronotum consists of small, fine punctures which contrast strongly with the widely spaced foveae on the mesonotum and propodeum. In opaca the pronotal sculpturing is composed of widely spaced foveae which are only slightly more dense than those on the mesonotum and propodeum. And in robynmae the sculpturing consists of small foveae on the pronotum which contrasts markedly with the widely spaced foveae on mesonotum and propodeum. In addition, the density of the sculpturing across the width of the propodeum is variable (weakest medially, stronger laterally) in kraepelini and robynmae and uniform in opaca. The shape of the petiolar node also differs across these species. It is narrowest and shortest in kraepelini, relatively longer and broader in opaca and long but narrow in robynmae. Essentially all presently known material can be unambiguously sorted into three sets representing these three taxa based on these character systems. In all other respects the material of these taxa is essentially identical or the differences are slight and random and show no obvious patterns. While kraepelini is allopatric to the others, opaca and robynmae have been collected together (from the same litter sample) in PNG.
The only apparent exception to this pattern is a single collection from Palolo, Palu, C. Celebes, Indonesia. In these specimens, the punctations on the propodeal dorsum are somewhat intermediate between kraepelini and opaca, although they are more similar to typical kraepelini than typical opaca. This is consistent with other material from Sulawesi which is typical of kraepelini. A reexamination of Brown’s Micronesian material has failed to uncover his “intergradient” forms as all could be placed with confidence into kraepelini, opaca or robynmae.
Keys including this Species
- Key to Amblyoponinae genera in Hong Kong SAR, China
- Key to Indo-Pacific Prionopelta Species
- Key to Micronesian Ants
Distribution
Shattuck (2008) - This is one of the most widely distributed species in the genus, being found from Sumatra and Peninsular Malaysia east through the Philippines and Micronesia to Samoa.
Latitudinal Distribution Pattern
Latitudinal Range: 32.4635° to -6.75°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Indo-Australian Region: Borneo, Fiji, Indonesia (type locality), Krakatau Islands, Malaysia, Micronesia (Federated States of), Palau, Philippines, Samoa, Singapore.
Oriental Region: India, Sri Lanka, Taiwan.
Palaearctic Region: China.
 Hamer et al. (2023), Fig. 2. Distribution map of Amblyoponinae species in Hong Kong (SAR), iNat = iNaturalist. A) Prionopelta kraepelini; B) Stigmatomma amblyops, S. crypticum, S. draconis; C) Stigmatomma luyiae, S. pagei, S. zwaluwenburgi; D) Stigmatomma nr. quadratum. Dark green zones indicate greater tree cover.
Hamer et al. (2023), Fig. 2. Distribution map of Amblyoponinae species in Hong Kong (SAR), iNat = iNaturalist. A) Prionopelta kraepelini; B) Stigmatomma amblyops, S. crypticum, S. draconis; C) Stigmatomma luyiae, S. pagei, S. zwaluwenburgi; D) Stigmatomma nr. quadratum. Dark green zones indicate greater tree cover.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
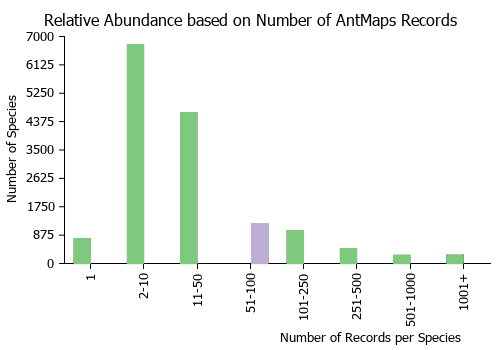
|
Biology
Hamer et al. (2023) - Leaf litter sampling within urban green spaces have resulted in numerous collections of P. kraepelini throughout Hong Kong. Urban sampling sites had little leaf litter hummus layer, and were largely composed of loose soil, stones and general anthropogenic debris. Sampling from more natural environments such as secondary forests, Feng Shui Woods and shrub lands, while commonly performed, has resulted in no specimens (Lee et al., 2021, Nooten et al., 2021). The collection of specimens from urban environments in Hong Kong reinforces the hypothesis that this species is indeed an exotic tramp species, being easily relocated via anthropogenic means (Ward 2006). In contrast, in Macau where recent surveys were performed and included leaf litter extractions, no P. kraepelini individuals have been detected to this date (Leong et al., 2017; Brassard et al., 2021).
The relative commonness of P. kraepelini in Hong Kong is interesting to note as this species is known only from a single collection in both Taiwan (Liuguei, Kaohsiung province, Terayama 2009) and Vietnam (Hanoi periphery, Anh et al., 2010), which represents the closest geographic records, but still located at 650 km and 850 km respectively from Hong Kong. While ecological information in Taiwan is missing, the record from Hanoi was collected from an organic Pomelo orchard, thus supporting the affinity of this species for disturbed habitats and exotic status in East Asia. Similarly, records from northern India were collected along roadsides and within fragmented habitats, but not in forested habitats (Bharti & Wachkoo 2012), while those from Sri Lanka appears to originate from a small forest fragment surrounded by urban development, based on Google Earth observations, with no ecological information provided directly in the original article (Dias et al., 2018). In contrast, other material collected from SE Asia appears to originate principally from forested habitats (Shattuck 2008) suggesting a native range that includes the Sundaland and Wallacean regions. Recent records from southern Thailand and collected in 2019 are from both secondary forests and oil palm plantation (Khachonpisitsak et al., 2020, Jaitrong pers. comm.), and the records from the Philippines did not include ecological information (General & Alpert 2012). Using Google Earth; specimens from the Philippines within Shattuck (2008) seemed to be recorded in both urban and agricultural habitats (MCZCT-215615), including a parkland (ANIC32-026204). It should be noted, however, that most of these specimens were collected within the last one hundred years, and the likelihood of environmental change since collection remains a possibility. While available information about the ecology of P. kraepelini is still fragmentary, habitat use observed for this species in several regions (Hong Kong, NW India, North Vietnam, Sri Lanka) suggest that it may have been introduced there, and that its native range may be limited to regions south of Peninsular Malaysia, the Philippines and the Wallacean region.
Castes
Worker
  
| |
| . | Owned by Museum of Comparative Zoology. |
Images from AntWeb
   
| |
| Worker. Specimen code casent0172314. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ANIC, Canberra, Australia. |
Male
Images from AntWeb
    
| |
| Male (alate). Specimen code casent0172315. Photographer April Nobile, uploaded by California Academy of Sciences. | Owned by ANIC, Canberra, Australia. |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- kraepelini. Prionopelta kraepelini Forel, 1905c: 3 (w.q.) INDONESIA (Java).
- Type-material: syntype workers, syntype queens (numbers not stated).
- Type-locality: Indonesia: Java, Buitenzorg (= Bogor), iii.1944 (K. Kraepelin).
- Type-depository: MHNG.
- Bharti & Wachkoo, 2012c: 818 (m.).
- Status as species: Emery, 1911d: 33; Chapman & Capco, 1951: 26; Brown, 1960a: 177, 220; Wilson & Taylor, 1967: 19; Terayama, 1989b: 343; Dlussky, 1994: 53; Bolton, 1995b: 365; Lin & Wu, 2003: 67; Wetterer & Vargo, 2003: 416; Clouse, 2007b: 259; Shattuck, 2008b: 23 (redescription); Terayama, 2009: 95; Zhou & Ran, 2010: 102; Pfeiffer, et al. 2011: 32 Bharti & Wachkoo, 2012c: 816 (redescription); Sarnat & Economo, 2012: 23; Bharti, Guénard, et al. 2016: 18.
- Distribution: Australia, Fiji Is, India, Indonesia (Java, Sulawesi), Malaysia (Peninsula, Sabah, Sarawak), Micronesia, Philippines (Luzon, Negros, Quezon), Samoa, Taiwan.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Hamer et al. (2023) - (n=9); HL 0.47-0.51; HW1 0.35-0.39; SL 0.24-0.27; ML 0.21-0.25; WL 0.55-0.61; DPW 0.18-0.21; DPL 0.14-0.17; T1W 0.3-0.35; GL 0.67-0.79; TL 2.29-2.58; CI 75.55-81.68; MI 41.91-49.21; SI 65.82-71.97; PI 125.15-145
Shattuck (2008) - Anterolateral corners of head, near mandibular insertions, rounded and lacking a tooth. Dorsal pronotal sculpturing consisting of fine punctations which contrast markedly with widely spaced foveae on mesonotum and propodeum. Foveae on dorsum of propodeum varying across its width (weakest medially, stronger laterally). Lateral mesosomal sculpturing consisting of small foveae on pronotum and anterior and ventral region of mesopleuron, dorsal region of mesopleuron and majority of propodeum smooth. Fenestra generally present but sometimes weakly developed within subpetiolar process. Colour pale yellow to yellow-red.
Queen
Hamer et al. (2023) - (alate) (n=1); HL 0.65-0.65; HW1 0.52-0.52; SL 0.35-0.35; ML 0.25-0.25; WL 0.87-0.87; DPW 0.27-0.27; DPL 0.16-0.16; T1W 0.4-0.4; GL 1-1; TL 3.2-3.2; CI 80.15-80.15; MI 39.08-39.08; SI 67.61-67.61; PI 170.37-170.37
Male
Hamer et al. (2023) - (n=10); HL 0.41-0.49; HW1 0.44-0.5; SL 0.14-0.16; ML 0.17-0.21; WL 0.66-0.78; DPW 0.15-0.2; DPL 0.11-0.18; T1W 0.26-0.33; GL 0.59-0.84 ; TL 2.16-2.66 ; CI 93.21-109.39; MI 37.42-48.09; SI 30-33.1; PI 82.44-140
Type Material
- Syntype, worker(s), queen(s), Tjompea, near Bogor (as Buitenzorg), Java, Indonesia, Musee d'Histoire Naturelle Genève.
References
- Bharti, H. & Wachkoo, A.A. 2012. First record of Prionopelta kraepelini (Hymenoptera: Formicidae) from India, with description of male caste. Sociobiology 59, 815-821.
- Forel, A. 1905f. Ameisen aus Java. Gesammelt von Prof. Karl Kraepelin 1904. Mitteilungen aus dem Naturhistorischen Museum in Hamburg 22: 1-26. (page 3, worker, queen described)
- Hamer, M.T., Pierce, M.P., Guénard, B. 2023. The Amblyoponinae (Formicidae) of Hong Kong. Asian Myrmecology 16, e016005 (doi:10.20362/am.016005).
- Khachonpisitsak, S., Yamane, S., Sriwichai, P., Jaitrong, W. 2020. An updated checklist of the ants of Thailand (Hymenoptera, Formicidae). ZooKeys 998, 1–182 (doi:10.3897/zookeys.998.54902).
- Shattuck, S.O. 2008b. Revision of the ant genus Prionopelta in the Indo-Pacific region. Zootaxa 1846:21-34.
- Wang, W.Y., Soh, E.J.Y., Yong, G.W.J., Wong, M.K.L., Benoit Guénard, Economo, E.P., Yamane, S. 2022. Remarkable diversity in a little red dot: a comprehensive checklist of known ant species in Singapore (Hymenoptera: Formicidae) with notes on ecology and taxonomy. Asian Myrmecology 15: e015006 (doi:10.20362/am.015006).
- Yamane, S., Tanaka, H.O., Hasimoto, Y., Ohashi, M., Meleng, P., Itioka, T. 2021. A list of ants from Lambir Hills National Park and its vicinity, with their biological information: Part II. Subfamilies Leptanillinae, Proceratiinae, Amblyoponinae, Ponerinae, Dorylinae, Dolichoderinae, Ectatomminae and Formicinae. Contributions from the Biological Laboratory, Kyoto University 31, 87–157.
References based on Global Ant Biodiversity Informatics
- Abe T., S. Yamane, and K. Onoyama. Ants collected on the Krakatau Islands 100 years after the great eruptions. Biogeography 14: 65-75.
- Anh L. N., K. Ogata, and S. Hosoichi. 2010. Ants of agricultural fields in Vietnam (Hymenoptera: Formicidae). Bull. Inst. Trop. Agr., Kyushu Univ. 33: 1-11.
- Bharti H., and A. A. Wachkoo. 2012. First record of Prionopelta kraepelini (Hymenoptera: Formicidae) from India, with description of male caste. Sociobiology 59: 815-821.
- Brown W. L., Jr. 1960. Contributions toward a reclassification of the Formicidae. III. Tribe Amblyoponini (Hymenoptera). Bulletin of the Museum of Comparative Zoology 122: 143-230.
- CSIRO Collection
- Chapman, J. W., and Capco, S. R. 1951. Check list of the ants (Hymenoptera: Formicidae) of Asia. Monogr. Inst. Sci. Technol. Manila 1: 1-327
- Chapman, J.W. and S.R. Capco. 1951. Check list of the ants (Hymenoptera: Formicidae) of Asia. Monographs of the Institute of Science and Technology (Manila) 1: 1- 327
- Emery C. 1911. Hymenoptera. Fam. Formicidae. Subfam. Ponerinae. Genera Insectorum 118: 1-125.
- Forel A. 1905. Ameisen aus Java. Gesammelt von Prof. Karl Kraepelin 1904. Mitt. Naturhist. Mus. Hambg. 22: 1-26.
- Guénard B., and R. R. Dunn. 2012. A checklist of the ants of China. Zootaxa 3558: 1-77.
- Hashimoto Y., and M. Mohamed. 2011. Ground-dwelling ant diversity in Maliau Basin, Borneo: evaluation of hand-sorting methods to estimate ant diversity. Tropics 19(2): 85-92.
- Ito, F.; Yamane, S.; Eguchi, K.; Noerdjito, W. A.; Kahono, S.; Tsuji, K.; Ohkawara, K.; Yamauchi, K.; Nishida, T.; Nakamura, K. 2001. Ant species diversity in the Bogor Botanic Garden, West Java, Indonesia, with descriptions of two new species of the genus Leptanilla (Hymenoptera, Formicidae). Tropics 10:379-404.
- Karavaiev V. 1926. Ameisen aus dem Indo-Australischen Gebiet. Treubia 8: 413-445.
- Li Z.h. 2006. List of Chinese Insects. Volume 4. Sun Yat-sen University Press
- Ngoc Anh L., K. Ogata, and S. Hosoishi. 2010. Ants of agricultural fields in Vietnam (Hymenoptera: Formicidae). Bull. Inst. Trop. Agr. Kyushu Univ. 33: 1-11.
- Pfeiffer M., and D. Mezger. 2012. Biodiversity Assessment in Incomplete Inventories: Leaf Litter Ant Communities in Several Types of Bornean Rain Forest. PLoS ONE 7(7): e40729. doi:10.1371/journal.pone.0040901
- Pfeiffer M.; Mezger, D.; Hosoishi, S.; Bakhtiar, E. Y.; Kohout, R. J. 2011. The Formicidae of Borneo (Insecta: Hymenoptera): a preliminary species list. Asian Myrmecology 4:9-58
- Shattuck, S. O. 2008. Revision of the ant genus Prionopelta (Hymenoptera: Formicidae) in the Indo-Pacific region. Zootaxa 1846:21-34.
- Terayama M. 1989. The ant tribe Amblyoponini (Hymenoptera, Formicidae) of Taiwan with description of a new species. Japanese Journal of Entomology 57:343-346.
- Terayama M. 2009. A synopsis of the family Formicidae of Taiwan (Insecta: Hymenoptera). Research Bulletin of Kanto Gakuen University. Liberal Arts 17:81-266.
- Terayama Mamoru. 2009. A synopsis of the family Formicidae of Taiwan (Insecta, Hymenoptera). The Research Bulletin of Kanto Gakuen University 17: 81-266.
- Wilson, Edward O. and Hunt, George L. Jr. 1967. Ant Fauna of Futuna and Wallis Islands, Stepping Stones to Polynesia. Pacific Insects. 9(4):563-584
- Woodcock P., D. P. Edwards, R. J. Newton, C. Vun Khen, S. H. Bottrell, and K. C. Hamer. 2013. Impacts of Intensive Logging on the Trophic Organisation of Ant Communities in a Biodiversity Hotspot. PLoS ONE 8(4): e60756. doi:10.1371/journal.pone.0060756
- Woodcock P., D. P. Edwards, T. M. Fayle, R. J. Newton, C. Vun Khen, S. H. Bottrell, and K. C. Hamer. 2011. The conservation value of South East Asia's highly degraded forests: evidence from leaf-litter ants. Phil. Trans. R. Soc. B. 366: 3256-3264.
- Yamane S. 2013. A Review of the ant fauna of the Krakatau Islands, Indonesia. Bull. Kitakyushu Mus. Nat. Hist. Hum. Hist. Ser: A, 11: 1-66


