Odontomachus caelatus
| Odontomachus caelatus | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Ponerinae |
| Tribe: | Ponerini |
| Genus: | Odontomachus |
| Species: | O. caelatus |
| Binomial name | |
| Odontomachus caelatus Brown, 1976 | |
All of the collections from Brasil, Ecuador, and Guyana are known to have been collected in the forest, and probably this species always lives in the wet forest. Nests collected by me were taken in rotten wood or under leaves in the soil at the base of a large tree. Probably it ranges widely in the Amazon Basin and the Guyanan forests. (Brown 1978)
Identification
Brown (1976) - A member of the Odontomachus haematodus group. The worker of this species is distinguished above all by its large size and the clear longitudinal striation of the gastric dorsum and part or all of the mesonotum. The swollen, sulcate mesonotum and the petiolar form are also distinctive.
Keys including this Species
Distribution
Latitudinal Distribution Pattern
Latitudinal Range: 4.088888889° to -64.23°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Neotropical Region: Brazil (type locality), Ecuador, French Guiana, Peru, Suriname.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
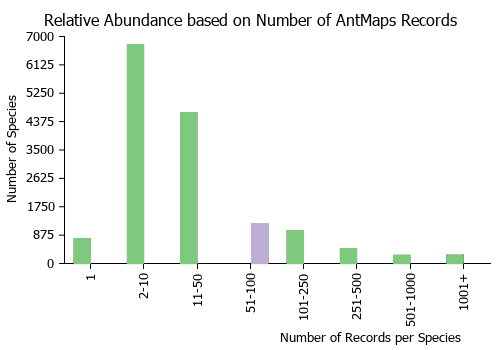
|
Biology
Castes
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- caelatus. Odontomachus caelatus Brown, 1976a: 140, fig. 23, pl. 1A (w.m.) BRAZIL (Amazonas, Amapá, Pará), PERU, ECUADOR, GUYANA.
- Type-material: holotype worker, 43 paratype workers.
- Type-locality: holotype Brazil: Amazonas State, Municipio of Manaus, Reserve
- a Ducke, 9.vi.1972, No. V-6, rotten wood, rainforest (W.L. & D.E. Brown); paratypes (number from each locality not stated): Brazil: Terr. Amapá, Serra do Navio (K. Lenko), Amazonas, Igarapé Marianil, on Rio Branco (Caracarai) Road, 20-24 km. NE Manaus, 22.viii.1962, No. M-7 (W.L. Brown), Pará, Pirelli Rubber Plantation, Iriboca, 21.viii.1962, No. B-279 (P.F. Darlington), nr mouth of R. Curuá, 30.iv.1963 (F.G. Werner), Peru: Dept. Loreto, viii.1939 (W.F. Walsh), Ecuador: Prov. Napo, Limoncocha (P.L. Kazan), Guyana: Great Batavia I., Cuyuni River, 4.viii.1920, Nos. 371, 374 (W.M. Wheeler), Kartabo, 16.vii.1920, No. 701 (W.M. Wheeler), Dunoon, 12.viii.1914, No. 340 (F.M. Gaige).
- Type-depositories: MZSP (holotype); MCZC, MZSP (paratypes).
- Status as species: Brandão, 1991: 362; Bolton, 1995b: 295; Rodriguez, J. 2008: 160; Bezděčková, et al. 2015: 124; Feitosa, 2015c: 99; Fernández & Guerrero, 2019: 538.
- Distribution: Brazil, Colombia, Ecuador, Guyana, Peru.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Holotype: TL 12.0, HL 3.20, HW (across vertex) 2.24, HW (across ocular prominences) 2.48, ML 1.66, scape L 2.94, eye L 0.47, WL 3.64 mm; Cl 70, Ml 52, SI 131.
Paratypes (smallest worker of 43, Great Batavia Island, Cuyuni R., Guyana; largest, Dept. Junin, Peru) TL 12.5-14.4, HL 3.24-3.76, HW (across vertex) 2.23-2.73, HW (across ocular prominences) 2.49-2.87, ML 1.70-1.92, scape L 3.04-3.33, eye L 0.48-0.55, WL 3.54-4.22; CI 69-73, MI 51-52, SI 122-136.
A large, robust species, reddish brown to dark brown in color, usually appearing black to the naked eye, with broad vertex and massive mandibles, resembling in a general way the large workers of Odontomachus bauri, Odontomachus laticeps and Odontomachus biumbonatus, and like them with palpi segmented 4,3. Diagnostic characters are mainly those of sculpture, pilosity-pubescence, and form of mesonotum and petiolar node.
(1) Dorsum of head and body very regularly and moderately finely striate (about 7 striae per 0.1 mm sample square on middle of left half of vertex), the striation extending far down the sides of the head, into the extraocular furrows and laterad of the eyes, and covering most of the temples. The furrows of the striation are microsculptured at the bottom, and their intervening ridges are only moderately shining, so that the general surface is moderately shining. Pronotal striation prevailingly longitudinal, becoming curved and generally trending concentrically around the edges of the disc. Mesonotum longitudinally striate in the middle, at least anteriorly ; longitudinal-arched, or even steeply arched on the sides, so that the posterior meso notal striae are obliquely diverging caudad, or in some cases, even transverse. Metanotal striation variable; longitudinal, oblique or transverse. Anterior tip of propodeum longitudinally, or else transversely striate like the rest of the propodeal dorsum. Petiolar node finely horizontally or obliquely striate around the front and sides on the lower 2/3 or so, the striation becoming indistinct and erratic higher up; apical spine nearly or quite smooth. Disc of first gastric segment evenly longitudinally striate, the furrows more or less distinctly septate or punctate, the striation becoming more broken and opaque on the sides of the tergum. Second segment also longitudinally striate, the furrows here more distinctly punctate. Third and fourth terga variably striate, fifth tergum nearly smooth, shining, with fine reticulate-striate or alutaceous sculpture on the sides. Mesepisternum usually completely striate, though the middle posterior part tends to have the striation weaker and more shining.
(2) Pilosity and pubescence reduced, except for a median pair of coarse suberect hairs at the usual position in the middle of the cranial dorsum, a few (1-9) coarse curved hairs on the pranotal disc, 0-4 on mesonotum, and 30-50 thick but pointed hairs, mostly 0.4-0.6 mm long, on dorsal surface of gaster; others 0.3-0.4 mm long on ventral surface. A few very fine, short hairs exist on the clypeus. Pubescence obsolescent, appressed to subappressed, detectable only in certain lights on the gastric dorsum, where on the first tergum the individual tiny hairs are spaced apart at an average distance about the same as, or more than, their average length (Plate 1, B); usually a little more abundant and closer together on the second tergum, but here still dilute. Appressed, fine, sparse pubescence on femoral apices, and becoming somewhat denser on tibiae, tarsi and antennae; absent or extremely inconspicuous on head, trunk and petiolar node.
(3) Mesonotum more strongly convex than usual in the genus, especially anteriad, but also more or less distinctly longitudinally sulcate in the middle.
(4) Transverse ridge between metasternal coxae at most forming a pair of low, blunt lobes with a hollow between.
(5) Petiole distinctive in form; above the short, weakly convex basal part, a long sloping section of the side-view profile of the node is nearly straight; profile of posterior face gently convex just above mid-height; apical spine slender, laterally compressed, usually slightly to distinctly back-tilted from the main axis of the node. Rear face of node weakly impressed near the middle.
(6) Color reddish brown (possibly faded) to blackish brown; appendages, especially antennal funiculi, sometimes a bit lighter and more reddish than rest of body. Bluish opalescence sometimes present on mandibles, antennal fossae, sides of head, etc., but less marked than in some other species.
Male
(a single example from the holotype nest series): TL 8.4, HL 1.24, HW (including eyes) 1.60, eye L 0.75, WL 3.48, forewing L 5.8 mm. Color dark brown; head and trunk almost black.
Mandibles about 0.22 mm long (insertion to apex); apices irregularly rounded; a diagonal carina runs from the outer margin near base to mesal margin near apex; mesal to this carina, nearly the basal half of the dorsal surface is whitish in color, concave, and probably semimembranous; this area may represent a holding and evaporating basin of mandibular gland pheromones. Maxillary palpi 5-merous, the apical segment very long and slender, and apparently consisting of 2 segments fused; labial palpi each 3-merous. Clypeus strongly convex.
Mesonotum finely longitudinally striate, subopaque; notauli (true parapsidal sutures) obsolete; scutum with a strong median longitudinal seam, carinate in front and groove-like behind; scutellum convex, with a median sulcus. Metanotum narrow, transversely striate, with a strong, raised, sharply rounded median carina. Propodeum with indistinct striation in front, diverging in a broad V behind, becoming transverse on the declivitous area. Petiole and gastric dorsum finely roughened, only feebly shining.
Erect hairs delicate, sparse, chiefly on mandibles, mesonotum and gaster. Body and appendages generally with a rather dense, short, brown appressed to suberect pubescence.
Distinguished from the males of Odontomachus haematodus and Odontomachus bauri by its larger size and uniformly dark color.
Type Material
Holotype worker (Museu de Zoologia da Universidade de Sao Paulo) and accompanying workers and a male from the same nest are from the Rcserva Ducke, Municipio of Manaus, Amazonas State, Brasil, W. L. and D. E. Brown, No. V-6, 9 June 1972, in rotten wood in rain forest. Paratypes (43 workers) are from widely separated localities in the Amazon-Guyana basins, all in rain forest: BRASIL: Terr. Amapa: Serra do Navio (K. Lenko). Amazonas: Igarape Marianil, on Rio Branco (Caracarai) Road, 20-24 km NE of Manaus (W. L. Brown, 22 Aug 1962, No. M-7). Para: Pirelli Rubber Plantation, Iriboca (P. F. Darlington, 21 Aug 1962, No. B-279. Near mouth of R. Curua, 30 April 1963 (F. G. Werner). PERU: Dept. Loreto, W. F. Walsh, Jr., Aug. 1939. ECUADOR: Provo Napo, Limoncocha (P. L. Kazan). GUYANA: Great Batavia Island, Cuyuni River (W. M. Wheeler, 4 Aug. 1920, Nos. 371, 374). Kartabo (Wheeler, 1 6 July 1920, No. 701). Dunoon (F. M. Gaige, 12 Aug. 1914, No. 340). Paratypes deposited in MZSP, Museum of Comparative Zoology, CU, and WWK.
References
- Albuquerque, E., Prado, L., Andrade-Silva, J., Siqueira, E., Sampaio, K., Alves, D., Brandão, C., Andrade, P., Feitosa, R., Koch, E., Delabie, J., Fernandes, I., Baccaro, F., Souza, J., Almeida, R., Silva, R. 2021. Ants of the State of Pará, Brazil: a historical and comprehensive dataset of a key biodiversity hotspot in the Amazon Basin. Zootaxa 5001, 1–83 (doi:10.11646/zootaxa.5001.1.1).
- Brown, W. L., Jr. 1976c. Contributions toward a reclassification of the Formicidae. Part VI. Ponerinae, tribe Ponerini, subtribe Odontomachiti. Section A. Introduction, subtribal characters. Genus Odontomachus. Stud. Entomol. 19: 67-171. (page 140, fig. 23, pl. 1A worker described)
- Camargo, K.S. de. 2011. Composicao e diversidade de "Poneromorfas" (Hymenoptera, Formicidae) em duas fitofisionomias de cerrado e padroes de distribuicao de "Poneromorfas", Pseudomyrmecinae e Cephalotini (Myrmicinae) para o Brasil. Thesis, Universidade de Brasilia.
- Franco, W., Ladino, N., Delabie, J.H.C., Dejean, A., Orivel, J., Fichaux, M., Groc, S., Leponce, M., Feitosa, R.M. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674, 509–543 (doi:10.11646/zootaxa.4674.5.2).
References based on Global Ant Biodiversity Informatics
- Brandao, C.R.F. 1991. Adendos ao catalogo abreviado das formigas da regiao neotropical (Hymenoptera: Formicidae). Rev. Bras. Entomol. 35: 319-412.
- Brown W. L., Jr. 1976. Contributions toward a reclassification of the Formicidae. Part VI. Ponerinae, tribe Ponerini, subtribe Odontomachiti. Section A. Introduction, subtribal characters. Genus Odontomachus. Stud. Entomol. 19: 67-171.
- Delabie J. H. C., R. Céréghino, S. Groc, A. Dejean, M. Gibernau, B. Corbara, and A. Dejean. 2009. Ants as biological indicators of Wayana Amerindian land use in French Guiana. Comptes Rendus Biologies 332(7): 673-684.
- Fernandes I., and J. de Souza. 2018. Dataset of long-term monitoring of ground-dwelling ants (Hymenoptera: Formicidae) in the influence areas of a hydroelectric power plant on the Madeira River in the Amazon Basin. Biodiversity Data Journal 6: e24375.
- Fernández F. 2008. Subfamilia Ponerinae s.str. Pp. 123-218 in: Jiménez, E.; Fernández, F.; Arias, T.M.; Lozano-Zambrano F. H. (eds.) 2008. Sistemática, biogeografía y conservación de las hormigas cazadoras de Colombia. Bogotá: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, xiv + 609 pp.
- Fernández F., and T. M. Arias-Penna. 2008. Las hormigas cazadoras en la región Neotropical. Pp. 3-39 in: Jiménez, E.; Fernández, F.; Arias, T.M.; Lozano-Zambrano, F. H. (eds.) 2008. Sistemática, biogeografía y conservación de las hormigas cazadoras de Colombia. Bogotá: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, xiv + 609 pp.
- Fernández, F. and S. Sendoya. 2004. Lista de las hormigas neotropicales. Biota Colombiana Volume 5, Number 1.
- Franco W., N. Ladino, J. H. C. Delabie, A. Dejean, J. Orivel, M. Fichaux, S. Groc, M. Leponce, and R. M. Feitosa. 2019. First checklist of the ants (Hymenoptera: Formicidae) of French Guiana. Zootaxa 4674(5): 509-543.
- Groc S., J. H. C. Delabie, F. Fernandez, M. Leponce, J. Orivel, R. Silvestre, Heraldo L. Vasconcelos, and A. Dejean. 2013. Leaf-litter ant communities (Hymenoptera: Formicidae) in a pristine Guianese rainforest: stable functional structure versus high species turnover. Myrmecological News 19: 43-51.
- Groc S., J. Orivel, A. Dejean, J. Martin, M. Etienne, B. Corbara, and J. H. C. Delabie. 2009. Baseline study of the leaf-litter ant fauna in a French Guianese forest. Insect Conservation and Diversity 2: 183-193.
- Scott-Santos, C.P., F.A. Esteves, C.R.F. Brandao. 2008. Catalogue of "Poneromorph" ant type specimens (Hymenoptera, Formicidae) deposited in the Museu de Zoologia da Universidade de Sao Paulo, Brazil. Papeis Avulsos de Zoologia 48(11):75-88.
- Vasconcelos, H.L., J.M.S. Vilhena, W.E. Magnusson and A.L.K.M. Albernaz. 2006. Long-term effects of forest fragmentation on Amazonian ant communities. Journal of Biogeography 33:1348-1356
- da Silva de Oliveira A. B., and F. A. Schmidt. 2019. Ant assemblages of Brazil nut trees Bertholletia excelsa in forest and pasture habitats in the Southwestern Brazilian Amazon. Biodiversity and Conservation 28(2): 329-344.
- de Souza J. L. P., F. B. Baccarob, V. L. Landeirob, E. Franklinc, and W. E. Magnussonc. 2012. Trade-offs between complementarity and redundancy in the use of different sampling techniques for ground-dwelling ant assemblages. Applied Soil Ecology 56: 63 73.

