Aenictus ceylonicus
| Aenictus ceylonicus | |
|---|---|

| |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Dorylinae |
| Genus: | Aenictus |
| Species: | A. ceylonicus |
| Binomial name | |
| Aenictus ceylonicus (Mayr, 1866) | |
| Synonyms | |
| |
| Common Name | |
|---|---|
| Chairo-himesasurai-ari | |
| Language: | Japanese |
Little is known about the biology of Aenictus ceylonicus.
Identification
A member of the ceylonicus group. Jaitrong and Yamane (2013) – Aenictus ceylonicus is most similar to Aenictus watanasiti in having the mandible with only 3 teeth including apical tooth. It can be separated from the latter by the postpetiole being short and clearly shorter than high (mandible with more than 3 teeth and postpetiole almost as long as high or longer in the other members of the group). See also under Aenictus formosensis.
Key to Aenictus species groups
Keys including this Species
Distribution
Aenictus ceylonicus is probably restricted to the western part of the Oriental region (India and Sri Lanka) (Jaitrong & Yamane, 2013). The Australian records shown on the occurrence map below are subspecies that have been raised to full species.
Latitudinal Distribution Pattern
Latitudinal Range: 32.6979° to -30.75°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Oriental Region: India (type locality), Sri Lanka (type locality).
Palaearctic Region: Japan.
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |

|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |

|
Biology
Castes
  
| |
| . | Owned by Museum of Comparative Zoology. |
| |
| . | |
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- ceylonicus. Typhlatta ceylonica Mayr, 1866a: 505 (w.) SRI LANKA.
- Type-material: lectotype worker (by designation of Jaitrong & Yamane, 2013: 220), 2 paralectotype workers.
- Type-locality: lectotype Sri Lanka (“Ceylon”): (no further data) (received from Sichel); paralectotypes with same data.
- Type-depository: NHMW (lectotype), MCZC, NHMW (paralectotypes).
- [Misspelled as ceylonensis by Wheeler, W.M. 1930a: 94, Chapman & Capco, 1951: 11; misspelled as ceylonicsu by Lin & Wu, 2003: 60.]
- Wheeler, G.C. & Wheeler, J. 1984: 265 (l.).
- Combination in Eciton (Aenictus): Karavaiev, 1926d: 422;
- combination in Aenictus: Dalla Torre, 1893: 7; Wilson, 1964a: 452.
- Status as species: Mayr, 1879: 668 (in key); Dalla Torre, 1893: 7; Forel, 1901a: 477; Bingham, 1903: 22; Forel, 1906b: 91; Emery, 1910b: 29; Wheeler, W.M. 1930a: 94, Menozzi, 1939a: 327; Chapman & Capco, 1951: 11; Wilson, 1964a: 452; Taylor & Brown, 1985: 52; Taylor, 1987a: 6; Wu, J. & Wang, 1992: 1303; Wang, M., 1992: 679; Xu, 1994a: 118; Bolton, 1995b: 59; Wu, J. & Wang, 1995: 50; Zhou & Chen, 1999: 63 (in key); Tiwari, 1999: 17; Zhou, 2001b: 59; Imai, et al. 2003: 209; Lin & Wu, 2003: 60; Jaitrong & Nabhitabhata, 2005: 11; Shattuck, 2008c: 16; Terayama, 2009: 124; Pfeiffer, et al. 2011: 32; Bharti, Wachkoo & Kumar, 2012: 293 (in key); Guénard & Dunn, 2012: 22; Jaitrong & Yamane, 2013: 220 (redescription); Bharti, Guénard, et al. 2016: 20; Dias, R.K.S. et al. 2020: 22.
- Senior synonym of latro: Wilson, 1964a: 452; Bolton, 1995b: 59; Zhou, 2001b: 59; Shattuck, 2008c: 16; Jaitrong & Yamane, 2013: 220.
- Distribution: India, Sri Lanka.
- [Note: Shattuck, 2008c: 16, restricted the distribution from the previous very wide distribution reported by Wilson, 1964a: 452.]
- latro. Aenictus ceylonicus var. latro Forel, 1901a: 477 (w.) INDIA (Maharashtra).
- Type-material: syntype workers (number not stated).
- [Note: Baroni Urbani, 1977e: 66, cites 3w syntypes NHMB.]
- Type-locality: India: Poona (R.C. Wroughton).
- Type-depositories: MHNG, NHMB.
- Subspecies of ceylonicus: Bingham, 1903: 22; Emery, 1910b: 29; Karavaiev, 1926d: 422; Chapman & Capco, 1951: 11.
- Junior synonym of ceylonicus: Wilson, 1964a: 452; Bolton, 1995b: 60; Zhou, 2001b: 59; Shattuck, 2008c: 16; Jaitrong & Yamane, 2013: 220.
Shattuck (2008) offers the following taxonomic notes on this species:
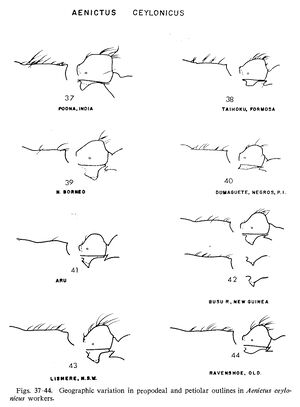
As previously conceived (Wilson, 1964: 452) this species extended from India and Sri Lanka eastward to Taiwan and south to Australia and contained eight junior synonyms (formosensis Forel, latro Forel, orientalis Karavaiev, papuanus Donisthorpe, similis Donisthorpe, and turneri Forel (with its junior synonyms deuqueti Crawley and exiguus Clark)). When discussing the specimens placed in ceylonicus Wilson (1964) recognised at least some of the variation noted in this study (for example, see Wilson’s figs. 37–44), but interpreted this variation as intraspecific. For example he mentioned that the subpetiolar process varies considerably in its development, but did not appreciate that this variation occurs in discrete states and shows a strong geographic pattern suggesting that a series of species are involved. A careful re-examination of these characters, combined with considerably more material, has resulted in significantly different conclusions being drawn compared to Wilson (1964).
An examination of currently available material has found that the old “ceylonicus” contains a large number of species, including Aenictus ceylonicus (strict sense), Aenictus acerbus, Aenictus orientalis, Aenictus papuanus, Aenictus prolixus and Aenictus turneri. To determine the identity of A. ceylonicus itself will require considerable work and is beyond the scope of the present study. However, there are a wealth of morphological characters which allow the development of robust species hypotheses as has been demonstrated above for the Australian fauna. Having said that, morphological differences among species are often subtle and require considerable attention to detail to decipher. The following notes are provided as a starting point for a full revision of these ants.
Most of the Indian specimens share the configuration of the subpetiolar process, which forms a rounded anterior lobe followed by a posterior flat to concave extension ending at the junction with the postpetiole. Others have an elongate rectangular subpetiolar process, including the types of A. latro. Specimens with both of these morphologies can be found throughout Asia including in Taiwan, the Philippines, Vietnam and Indonesia. But while material from Vietnam has a rectangular subpetiolar process it has the dorsal surface of the mesosoma smooth and lacking any indication of the metanotal groove (most other species have at least a weak angle at the metanotal groove). Thus while the shape of the subpetiolar process is important it must be used in conjunction with other characters when determining species boundaries.
While the work undertaken here is preliminary, it clearly shows that the situation surrounding this species, and close relatives, is much more complex than that recognised by earlier workers. As a first step in clarifying this situation the names A. orientalis and A. turneri are treated as valid species, A. papuanus and A. similis are transferred to synonymy with A. orientalis while A. formosensis and A. latro are retained as junior synonyms of A. ceylonicus. However this should be treated as preliminary until all relevant material can be studied in detail.
Description
Worker
Jaitrong and Yamane (2013) - (lectotype and paralectotypes of A. ceylonicus, n = 3). TL 3.15–3.20 mm; HL 0.63–0.70 mm; HW 0.58–0.63 mm; SL 0.48–0.53 mm; ML 1.00–1.03 mm; PL 0.25–0.28 mm CI 89–92; SI 83–84. (lectotype and paralectotypes of A. ceylonicus).
Head in full-face view subrectangular, slightly longer than broad, sides convex and posterior margin almost straight or feebly concave. Antennal scape reaching 2/3 of head length; antennal segments II longer than broad and slightly longer than each of III-VI; III slightly longer than broad; IV-VI each almost as long as broad; terminal segment (X) almost as long as VII+VIII+IX. Frontal carina short, reaching the level of posterior margin of torulus. Parafrontal ridge absent. Anterior clypeal margin weakly concave, lacking denticles. Masticatory margin of mandible with 3 teeth, including a large apical tooth; basal margin weakly concave, lacking denticles. Maximum width of gap between anterior clypeal margin and mandibles about 1.8 times as broad as maximum width of mandible. Promesonotum convex dorsally and sloping gradually to metanotal groove; metanotal groove indistinct; metapleural gland bulla relatively large, its maximum diameter about 3.1 times as long as distance between propodeal spiracle and metapleural gland bulla. Propodeum in profile with feebly convex dorsal outline; propodeal junction angulated, nearly right-angled; declivity of propodeum wide, shallowly concave, and encircled with a distinct rim. Petiole sessile and short, almost as long as high, with its dorsal outline elevated posteriorly; subpetiolar process weakly developed. Postpetiole distinctly shorter than petiole and shorter than high, with its dorsal outline slightly elevated posteriorly.
Head entirely smooth and shiny. Mandible with fine longitudinal striation, except along masticatory margin smooth and shiny. Antennal scape smooth and shiny. Promesonotum smooth and shiny except anteriormost portion punctate; mesopleuron, metapleuron and propodeum with several longitudinal rugae. Lateral faces of petiole and postpetiole microreticulate but the dorsal faces smooth and shiny. Legs smooth and shiny.
Head and mesosoma dorsally with relatively sparse standing hairs; longest pronotal hair 0.15–0.18 mm long. Head, mesosoma, petiole and postpetiole reddish brown; gaster, antennae and legs yellowish brown, paler than head, mesosoma and waist.
Wilson (1964) - Syntypes: HW 0.62 mm, HL 0.62 mm, SL 0.50 mm; HW 0.54 mm, HL 0.60 mm, SL 0.44 mm. Antenna 10-segmented. Mandible narrow, bearing 1 large apical and 1 large preapical tooth; the basal portion right-angulate, which can be interpreted as a 3rd "basal" tooth. When mandibles are closed there remains a gap between their posterior borders and center of anterior clypeal border almost as wide as maximum width of mandible. Clypeus emarginate, not armed. Parafrontal ridge rudimentary. Occiput very feebly convex, lacking a collar. Propodeal junction surmounted by low, rounded, transverse ridge, which in side YIGW appears as a "rounded" right angle. Subpetiolar process a long, thin, evenly rounded lobe which projects anteriorly. Pilosity abundant and relatively long; length of longest hairs on pronotum about 0.16 mm.
Head entirely shining. Pronotum shining. Mesothorax and propodeum bearing 15-20 longitudinal rugae, their interspaces weakly microreticulate and feebly shining. Sides of petiole and postpetiole microreticulate and subopaque, their tops shining. Concolorous clear yellow.
Type Material
Jaitrong and Yamane (2013) – Typhlatta ceylonica: Three syntype workers from Sri Lanka (NHMW, examined). One worker among them is selected as the lectotype (Fig. 27E). Aenictus ceylonicus var. latro: Six syntype workers from India, Poona (MHNG, examined).
- Typhlatta ceylonica: Lectotype (designated by Jaitrong & Yamane, 2013), worker, Sri Lanka, Naturhistorisches Museum Wien, Vienna.
- Typhlatta ceylonica: Paralectotype (designated by Jaitrong & Yamane, 2013), 2 workers, Sri Lanka, Naturhistorisches Museum Wien, Vienna.
- Aenictus ceylonicus var. latro: Syntype, 6 workers, Poona, India, Musee d'Histoire Naturelle Genève.
- Aenictus ceylonicus var. latro: Syntype, 3 workers, Poona, India, Museum of Comparative Zoology.
References
- Antony, A.K., Prasad, G. 2022. Two new species of army ants of the Aenictus ceylonicus group (Hymenoptera: Formicidae) from Kerala, India. Journal of Threatened Taxa 14(3): 20780-20785 (doi:10.11609/jott.6962.14.3.20780-20785).
- Dalla Torre, K. W. von. 1893. Catalogus Hymenopterorum hucusque descriptorum systematicus et synonymicus. Vol. 7. Formicidae (Heterogyna). Leipzig: W. Engelmann, 289 pp. (page 7, Combination in Aenictus)
- Dhadwal, T., Bharti, H. 2023. Aenictus dirangensis sp. nov. (Hymenoptera: Formicidae), a new species of Aenictus ceylonicus group from India. Journal of the Entomological Research Society 25(2): 387-403 (doi:10.51963/jers.v25i2.2367).
- Jaitrong, W. & Yamane, S. 2013. The Aenictus ceylonicus species group (Hymenoptera, Formicidae, Aenictinae) from Southeast Asia. Journal of Hymenoptera Research 31:165-233.
- Khachonpisitsak, S., Yamane, S., Sriwichai, P., Jaitrong, W. 2020. An updated checklist of the ants of Thailand (Hymenoptera, Formicidae). ZooKeys 998, 1–182 (doi:10.3897/zookeys.998.54902).
- Liu, C., Fischer, G., Hita Garcia, F., Yamane, S., Liu, Q., Peng, Y.Q., Economo, E.P., Guénard, B., Pierce, N.E. 2020. Ants of the Hengduan Mountains: a new altitudinal survey and updated checklist for Yunnan Province highlight an understudied insect biodiversity hotspot. ZooKeys 978, 1–171 (doi:10.3897/zookeys.978.55767).
- Mayr, G. 1866a. Myrmecologische Beiträge. Sitzungsber. Kais. Akad. Wiss. Wien Math.-Naturwiss. Cl. Abt. I 53: 484-517 (page 505, worker described)
- Jaitrong, W. & Yamane, S. 2011. Synopsis of Aenictus species groups and revision of the A. currax and A. laeviceps groups in the eastern Oriental, Indo-Australian, and Australasian regions (Hymenoptera: Formicidae: Aenictinae). Zootaxa, 3128, 1–46. PDF
- Sahoo, B., Ramakrishnaiah, S., Rajan, P.D., Datta-Roy, A. 2023. A new species of army ant genus Aenictus (Hymenoptera: Formicidae) from India. Journal of the Entomological Research Society 25(3), 615-622 (doi:10.51963/jers.v25i3.2504).
- Shattuck, S.O. (2008) Review of the ant genus Aenictus (Hymenoptera: Formicidae) in Australia with notes on A. ceylonicus (Mayr). Zootaxa 1926, 1–19.
- Wheeler, G. C.; Wheeler, J. 1984a. The larvae of the army ants (Hymenoptera: Formicidae): a revision. J. Kans. Entomol. Soc. 57: 263-275 (page 265, larva described)
- Wilson, E. O. 1964a. The true army ants of the Indo-Australian area (Hymenoptera: Formicidae: Dorylinae). Pac. Insects 6: 427-483 (page 452, senior synonym of formosensis, latro, orientalis, papuanus, similis and turneri (and its junior synonyms deuqueti and exiguus))
- Wu, J. & Wang, C. 1992. Formicidae (pp. 1301-1320). In Peng, J. et al. Iconography of Forest Insects in Hunan, China. Forest Bureau of Hunan Province: 1473 pp. Hunan Scientific and Technical Publishing House.
- Yamane, S., Tanaka, H.O., Hasimoto, Y., Ohashi, M., Meleng, P., Itioka, T. 2021. A list of ants from Lambir Hills National Park and its vicinity, with their biological information: Part II. Subfamilies Leptanillinae, Proceratiinae, Amblyoponinae, Ponerinae, Dorylinae, Dolichoderinae, Ectatomminae and Formicinae. Contributions from the Biological Laboratory, Kyoto University 31, 87–157.
References based on Global Ant Biodiversity Informatics
- Bharti H., A. A. Wachkoo, and R. Kumar. 2017. First inventory of ants (Hymenoptera: Formicidae) in Northwestern Shivalik, India. Halteres 8: 33-68.
- Bui T.V. 2002. Result of ant survey in Tam Dao National Park. Hoi Nghi Con Trung Hoc Toan Quoc, Ha Noi 495-498.
- CSIRO Collection
- Chapman J. W. 1965. Studies on the ecology of the army ants of the Philippines genus Aenictus Schuckard (Hymenoptera: Formicidae). Philippine Journal of Science. 93: 551-595.
- Chapman J. W. 1963. Some new and interesting Philippine ants (Hymenoptera: Formicidae). Philipp. J. Sci. 92: 247-263.
- Chapman, J. W., and Capco, S. R. 1951. Check list of the ants (Hymenoptera: Formicidae) of Asia. Monogr. Inst. Sci. Technol. Manila 1: 1-327
- Chapman, J.W. and S.R. Capco. 1951. Check list of the ants (Hymenoptera: Formicidae) of Asia. Monographs of the Institute of Science and Technology (Manila) 1: 1- 327
- Chen P., Y. Su, S. S. Rao, Y. F. Long, and C. H. Du. 2012. Study on the ant diversity in different Illicium verum stands. Journal of West China Forestry Science 41(1): 60-68.
- Chen Y. Q., Q. Li, Y. L. Chen, Z. X. Lu, X. Y. Zhou. 2011. Ant diversity and bio-indicators in land management of lac insect agroecosystem in Southwestern China. Biodivers. Conserv. 20: 3017-3038.
- Dias R. K. S. 2002. Current knowledge on ants of Sri Lanka. ANeT Newsletter 4: 17- 21.
- Dias R. K. S. 2006. Current taxonomic status of ants (Hymenoptera: Formicidae) in Sri Lanka. The Fauna of Sri Lanka: 43-52. Bambaradeniya, C.N.B. (Editor), 2006. Fauna of Sri Lanka: Status of Taxonomy, Research and Conservation. The World Conservation Union, Colombo, Sri Lanka & Government of Sri Lanka. viii + 308pp.
- Dias R. K. S. 2013. Diversity and importance of soil-dweeling ants. Proceedings of the National Symposium on Soil Biodiversity, chapt 4, pp 19-22.
- Dias R. K. S., K. R. K. A. Kosgamage, and H. A. W. S. Peiris. 2012. The Taxonomy and Conservation Status of Ants (Order: Hymenoptera, Family: Formicidae) in Sri Lanka. In: The National Red List 2012 of Sri Lanka; Conservation Status of the Fauna and Flora. Weerakoon, D.K. & S. Wijesundara Eds., Ministry of Environment, Colombo, Sri Lanka. p11-19.
- Donisthorpe H. 1941. Descriptions of new species of ants from New Guinea. Annals and Magazine of Natural History (11)7: 129-144.
- Donisthorpe H. 1948. A fourth instalment of the Ross Collection of ants from New Guinea. Annals and Magazine of Natural History (12)1: 131-143.
- Emery C. 1910. Hymenoptera. Fam. Formicidae. Subfam. Dorylinae. Genera Insectorum 102: 1-34.
- Forel A. 1901. Les Formicides de l'Empire des Indes et de Ceylan. Part VIII. J. Bombay Nat. Hist. Soc. 13: 462-477
- Forel A. 1913. H. Sauter's Formosa-Ausbeute: Formicidae II. Arch. Naturgesch. (A)79(6): 183-202
- Guénard B., and R. R. Dunn. 2012. A checklist of the ants of China. Zootaxa 3558: 1-77.
- Heterick B. E., B. Durrant, and N. R. Gunawardene. 2010. The ant fauna of the Pilbara Bioregion, Western Australia. Records of the Western Australian Museum, Supplement 78: 157-167.
- Hua Li-zhong. 2006. List of Chinese insects Vol. IV. Pages 262-273. Sun Yat-sen university Press, Guangzhou. 539 pages.
- Jaitrong W., and S. Yamane. 2013. The Aenictus ceylonicus species group (Hymenoptera, Formicidae, Aenictinae) from Southeast Asia. Journal of Hymenoptera Research 31: 165-233.
- Janda M., G. D. Alpert, M. L. Borowiec, E. P. Economo, P. Klimes, E. Sarnat, and S. O. Shattuck. 2011. Cheklist of ants described and recorded from New Guinea and associated islands. Available on http://www.newguineants.org/. Accessed on 24th Feb. 2011.
- Li Z.h. 2006. List of Chinese Insects. Volume 4. Sun Yat-sen University Press
- Pfeiffer M.; Mezger, D.; Hosoishi, S.; Bakhtiar, E. Y.; Kohout, R. J. 2011. The Formicidae of Borneo (Insecta: Hymenoptera): a preliminary species list. Asian Myrmecology 4:9-58
- Poonjampa R., and D. Wiwatwitaya. 2001. Using ants as indicators of plant communities at Khao Yai National Park. Thai Journal of Forestry (19-21): 84-95.
- Robson Simon Ant Collection, 05-Sept-2014
- Shattuck, S. O. 2008. Review of the ant genus Aenictus (Hymenoptera: Formicidae) in Australia with notes on A. ceylonicus (Mayr). Zootaxa 1926:1-19.
- Sheela S. 2008. Handbook of Hymenoptera, Formicidae. Zoological Survey of India, 56 pages
- Song Y., Z Xu, C Li, N. Zhang, L. Zhang, H. Jiang, and F Mo. 2013. An analysis on the ant fauna of the Nangun River Nature Reserve in Yunnan, China. Forest Research 26(6): 773-780.
- Song Y., Z. Xu, C. Li, N. Zhang, L. Zhang, H. Jiang, and F. Mo. 2013. An Analysis on the Ant Fauna of the Nangun river Nature Reserve in Yunnan, China. Forest Research 26(6): 773-780.
- Suriyapong Y. 2003. Study of ground dwelling ant populations and relationship to some ecological factors in Sakaerat environmental research station, Nakhon Ratchasima. PhD thesis Suranaree University of Technology 203 pages.
- Taylor R. W. 1987. A checklist of the ants of Australia, New Caledonia and New Zealand (Hymenoptera: Formicidae). CSIRO (Commonwealth Scientific and Industrial Research Organization) Division of Entomology Report 41: 1-92.
- Taylor R. W., and D. R. Brown. 1985. Formicoidea. Zoological Catalogue of Australia 2: 1-149.
- Terayama M. 2000. A list of Japanese ants changed after "A guide for the identification of Japanase ants I, II, III" (2). Ari 24: 13-21.
- Terayama M. 2009. A synopsis of the family Formicidae of Taiwan (Insecta: Hymenoptera). Research Bulletin of Kanto Gakuen University. Liberal Arts 17:81-266.
- Terayama, M. 2009. A synopsis of the family Formicidae of Taiwan (Insecta; Hymenoptera). The Research Bulletin of Kanto Gakuen University 17: 81-266.
- Terayama. M. and Inoue. N. 1988. Ants collected by the members of the Soil Zoological Expedition to Taiwan. ARI Reports of the Myrmecologists Society (Japan) 18: 25-28
- Tiwari R. N. 1999. Taxonomic studies on ants of southern India (Insecta: Hymenoptera: Formicidae). Memoirs of the Zoological Survey of India 18(4): 1-96.
- Tiwari, R.N. 1999. Taxonomic studies on ants of southern India (Insecta: Hymenoptera: Formicidae). Memoirs of the Zoological Survey of India 18(4):1-96
- Varghese T. 2004. Taxonomic studies on ant genera of the Indian Institute of Science campus with notes on their nesting habits. Pp. 485-502 in : Rajmohana, K.; Sudheer, K.; Girish Kumar, P.; Santhosh, S. (eds.) 2004. Perspectives on biosystematics and biodiversity. Prof. T.C. Narendran commemoration volume. Kerala: Systematic Entomology Research Scholars Association, xxii + 666 pp.
- Wheeler W. M. 1929. Ants collected by Professor F. Silvestri in Formosa, the Malay Peninsula and the Philippines. Bollettino del Laboratorio di Zoologia Generale e Agraria della Reale Scuola Superiore d'Agricoltura. Portici 24: 27-64.
- Wheeler W. M. 1930. Formosan ants collected by Dr. R. Takahashi. Proceedings of the New England Zoological Club 11: 93-106.
- Wilson E. O. 1964. The true army ants of the Indo-Australian area (Hymenoptera: Formicidae: Dorylinae). Pacific Insects 6: 427-483.
- Xu Z. 1994. A taxonomic study of the ant subfamily Dorylinae China (Hymenoptera Formicidae Ponerinae). Journal of Southwest Forestry College 14(2): 115-122
- Yamane S.; Ikudome, S.; Terayama, M. 1999. Identification guide to the Aculeata of the Nansei Islands, Japan. Sapporo: Hokkaido University Press, xii + 831 pp. pp138-317.
- Zhang R. J., L. W. Liang, and S. Y. Zhou. 2014. An analysis on the ant fauna of Nonggang Nature Reserve in Guangxi, China. Journal of Guangxi Normal university: Natural Science Edition 32(3): 86-93.
