Ponera szentivanyi
| Ponera szentivanyi | |
|---|---|
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Formicidae |
| Subfamily: | Ponerinae |
| Tribe: | Ponerini |
| Genus: | Ponera |
| Species: | P. szentivanyi |
| Binomial name | |
| Ponera szentivanyi Wilson, 1957 | |
Only known from two type specimens.
Identification
Taylor (1967) - The 4-segmented club and lack of an incised mesometanotal suture allow placement to the tenuis group.
Differs from Ponera petila by its larger size, narrower head (CI 75 versus 78), and other dimensional differences. Sculpturation of the head and mesosoma of szentivanyi is about intermediate between that described for petila and szaboi.
Differs from Ponera szaboi by its smaller size (HW 0.30-0.31 mm, PW 0.23 mm, DPW 0.15 mm; opposed to 0.34 mm, 0.28 mm and 0.21-0.24 mm respectively in szentivanyi), relatively short scapes (SI 79-83, against 94 in szentivanyi) and a less intense sculpturation of the mesosoma.
Keys including this Species
Distribution
Known only from SE New Guinea.
Latitudinal Distribution Pattern
Latitudinal Range: -5.333° to -9.166667°.
| North Temperate |
North Subtropical |
Tropical | South Subtropical |
South Temperate |
- Source: AntMaps
Distribution based on Regional Taxon Lists
Indo-Australian Region: New Guinea (type locality).
Distribution based on AntMaps
Distribution based on AntWeb specimens
Check data from AntWeb
Countries Occupied
| Number of countries occupied by this species based on AntWiki Regional Taxon Lists. In general, fewer countries occupied indicates a narrower range, while more countries indicates a more widespread species. |
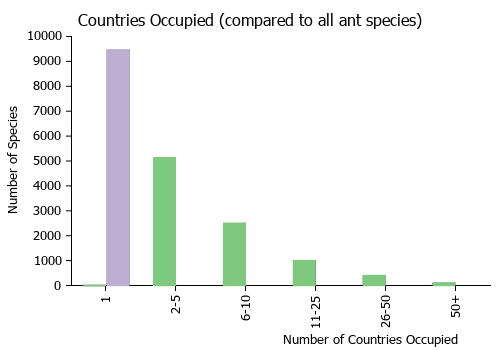
|
Estimated Abundance
| Relative abundance based on number of AntMaps records per species (this species within the purple bar). Fewer records (to the left) indicates a less abundant/encountered species while more records (to the right) indicates more abundant/encountered species. |
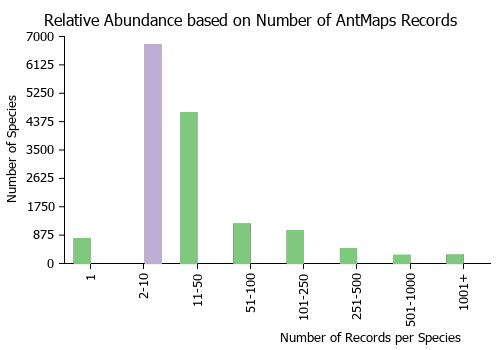
|
Biology
Nothing is known about the biology of Ponera szentivanyi.
|
Castes
Queen, male, and immature stages unknown.
Nomenclature
The following information is derived from Barry Bolton's Online Catalogue of the Ants of the World.
- szentivanyi. Ponera szentivanyi Wilson, 1957b: 372, figs. 1, 2 (w.) NEW GUINEA. See also: Taylor, 1967a: 103.
Unless otherwise noted the text for the remainder of this section is reported from the publication that includes the original description.
Description
Worker
Holotype. HW 0.34 mm; HL 0.45 mm, SL 0.32 mm, CI 76, 81 94, PW 0.28 mm (petiole height not measured; see paratype), petiolar node length 0.16 mm, dorsal petiole width 0.24 mm. Three well developed teeth occupying the apical two-fifths of the masticatory border, followed basally by an indeterminate number of minute denticles. Eye minute, consisting of a single ommatidium, located about 0.8 the distance from the lateral occipital border to the midpoint of the anterior genal border. Antennal club distinctly 4-jointed, considerably longer than the remainder of the funiculus. Head elongate (CI 76) with very feebly convex sides, and feebly but distinctly concave occipital border. Petiolar node seen from side relatively thin, elongate-trapezoidal; seen from directly above, so that the posterior face is exactly parallel with the line of vision, the node as a whole forms slightly more than a half-circle, but the dorsal surface alone forms much less than a half-circle; seen from the preceding position the posterior face is feebly but distinctly concave. Subpetiolar process somewhat reduced, its apex right-angular.
Body sculpturing approximately as described for P. huonica Wilson.
Pilosity and pubescence as in P. caledonica Wilson.
Body uniformly light brownish yellow; appendages clear yellow.
Paratype. A single callow worker taken with the holotype has the integument of the head somewhat crumpled and distorted through drying, so that regular cephalic measurements could not be made. PW 0.28 mm, petiole height 0.29 mm, dorsal petiole width 0.21 mm. Body color clear, pale yellow.
Taylor (1967) - Mesometanotal suture lacking on mesosomal dorsum. Palpal formula: Maxillary 2: Labial 2 (paratype inspected). Dimensions of holotype (listed first) and the single paratype are: HW 0.34mm; SL 0.32 mm; CI 75; SI 94; PW 0.28 mm; PNL 0.16 mm; PH 0.29 mm; DPW 0.24 mm, 0.21 mm; PNI 86, 75.
Type Material
PAPUA: Karema, near the Brown River, about 30 miles north of Port Moresby; March 8-11, 1955; Holotype and single paratype worker (Wilson, acc. no. 563). Museum of Comparative Zoology
Etymology
This species is named in honor of Dr. J. H. Szent-Ivany, the expert resident entomologist of the Territory of Papua-New Guinea, whose friendly assistance greatly aided the author's field work in this area.
References
- Csősz, S., Loss, A. C., Fisher, B. L. 2023. Exploring the diversity of the Malagasy Ponera (Hymenoptera: Formicidae) fauna via integrative taxonomy. Organisms Diversity & Evolution (doi:10.1007/s13127-023-00610-1).
- Taylor, R. W. 1967a. A monographic revision of the ant genus Ponera Latreille (Hymenoptera: Formicidae). Pac. Insects Monogr. 13: 1-112 (page 103, see also)
- Wilson, E. O. 1957b. The tenuis and selenophora groups of the ant genus Ponera (Hymenoptera: Formicidae). Bulletin of the Museum of Comparative Zoology 116: 355-386 (page 372, figs. 1, 2 worker described)
References based on Global Ant Biodiversity Informatics
- Janda M., G. D. Alpert, M. L. Borowiec, E. P. Economo, P. Klimes, E. Sarnat, and S. O. Shattuck. 2011. Cheklist of ants described and recorded from New Guinea and associated islands. Available on http://www.newguineants.org/. Accessed on 24th Feb. 2011.
- Taylor R. W. 1967. A monographic revision of the ant genus Ponera Latreille (Hymenoptera: Formicidae). Pacific Insects Monograph 13: 1-112.
- Wilson E. O. 1957. The tenuis and selenophora groups of the ant genus Ponera (Hymenoptera: Formicidae). Bulletin of the Museum of Comparative Zoology 116: 355-386.
- Wilson E. O. 1958. Studies on the ant fauna of Melanesia III. Rhytidoponera in western Melanesia and the Moluccas. IV. The tribe Ponerini. Bulletin of the Museum of Comparative Zoology 119: 303-371.
- Wilson Edward O. 1959. Adaptive Shift and Dispersal in a Tropical Ant Fauna. Evolution 13(1): 122-144